She has tried multiple times,
yet her attempts at losing weight and maintaining it have been unsuccessful
She is worried about her excess weight and risk of obesity-related complications*
She is starting to feel the physical and psychological consequences of excess weight in her daily life
See how people like Anjali may achieve
their weight goals with Mounjaro®
Get started *Hypothetical patient profile

Mounjaro is the first and only GIP and GLP-1 receptor agonist approved for weight management that may help patients living with obesity achieve their weight goals.1-3,*
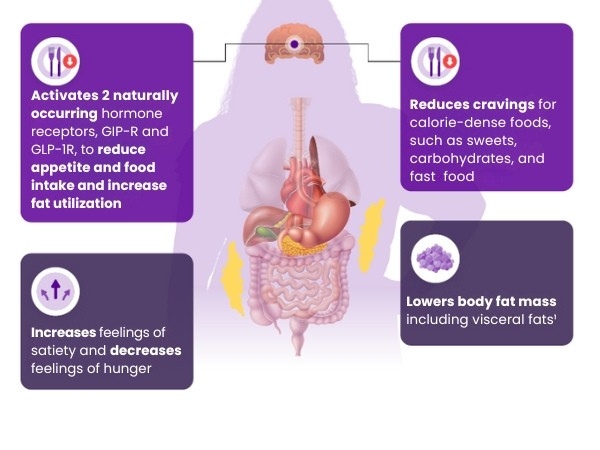
The role of GIP and GLP-1 receptors
- GIP and GLP-1 receptors are present in many body areas, including brain regions that regulate appetite1,a
- GIP receptors are present in adipose tissue and help regulate fat storage1,4
- GLP-1 regulates gastric emptying and feelings of satiety in the brain1,8
Brain
GIP activity8,b
Reduced food intake
GLP-1 activity6,8
Reduced food intake
Increased satiety
Subcutaneous
white adipose tissue
GIP activity4,8
Increased insulin sensitivity
Pancreas
GIP activity9
Increased insulin secretion
Increased glucagon in a glucose-dependent way
GLP-1 activity9
Increased insulin secretion
Reduced glucagon secretion
Stomach
GLP-1 activity7
Delayed gastric emptying

Mounjaro may have several effects on the body that help
support weight reduction in patients living with obesity1,10: