- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Improving Diversity in Clinical Trials: The Way Forward
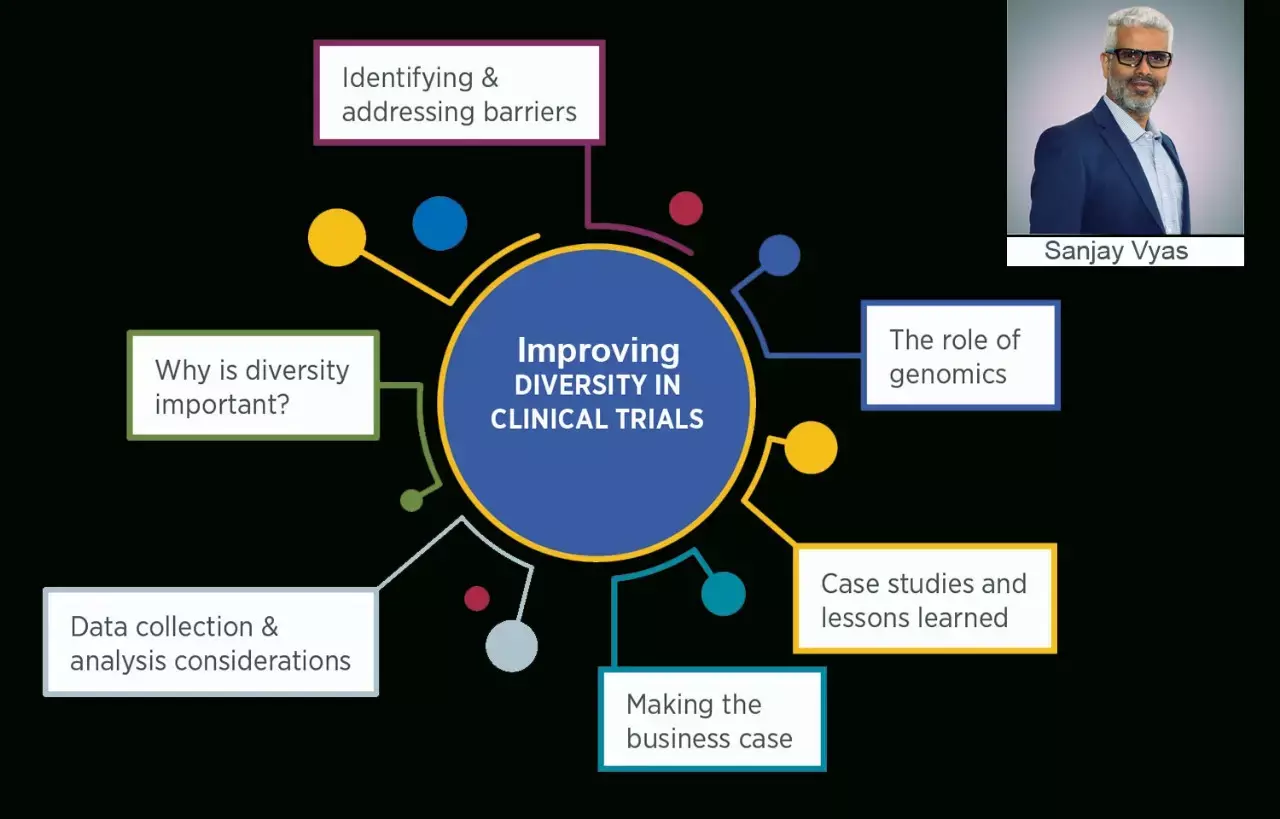
Historically, clinical trials have witnessed under-representation from participants of diverse races and ethnicity due to a variety of factors and barriers. Earlier this year, Parexel released our Discussions on Diversity report, a combination of qualitative and quantitative data, which identifies some of those critical barriers while offering specific solutions to help ensure more equitable access. The study, conducted across 10 countries, highlights that feeling of mistrust and skepticism by participants, insufficient translation of trial information materials in participant's primary language, financial challenges, childcare needs, and degree of stigma around illness in some communities, are among the leading causes of poor representation of diverse populations.
The Need for Diverse and Inclusive Clinical Research
The COVID-19 pandemic has had a significant impact on the CRO industry, affecting every aspect of the clinical trial continuity, from patient care and patient recruitment to site access and monitoring. However, the pandemic also opened numerous opportunities for innovation that demanded agile decision-making from drug developers. Deployment of digital technology and redirecting resources to pivot from the traditional way of conducting trials helped sustain the CRO industry during these tough times.
The basic assumption of clinical research is that investigators take data from a relatively small but representative sample and generalize the outcome to the larger patient population. However, if the sample is too uniform or constricted, it will hamper the broad applicability of the results. It is well-known that a patient's reactions, response, and outcomes to treatments vary by age, weight, sex, race, ethnicity, and other factors. Under-representation in clinical trials can result in biased, non-compliant outcomes that could be ineffective or sometimes dangerous.
Our society is diverse which demands that our research take into consideration that every individual and their characteristics are unique. These attributes must be considered and incorporated to ensure better clinical outcomes and reduce health and scientific risk.
Ways to Increase Diversity and Inclusion
Overcoming sex, racial, and cultural disparities in clinical trials can be minimized. Here's how:
Employ technology and data as drivers of diversity: Incorporating the latest technologies like wearables, cloud computing, artificial intelligence, and even advanced analytics in the right way can accelerate clinical trial outcomes and increase the chance of market success. Utilizing a synthetic control arm in place of a traditional control group, so that patients don't have to face the possibility of being randomized to a placebo control or to another control that might not be as effective is another way of improving efficiency. The synthetic control arms additionally accelerate the study timelines, allowing the FDA submissions to proceed more quickly.
Reduce logistical challenges: Decentralized and hybrid trial models that allow the trials to continue remotely can reduce major hurdles related to clinical trial participation logistics. For example, traditional, site-based trials often take place at large hospitals and academic instructions in major cities and require hours multiple times a month of the participants' time. These approaches are expensive, time-consuming, and burdensome for the average person. However, decentralized approaches utilize telehealth visits, direct-to-patient drug shipments, sensors, variable devices, home health nurse visits, and even remote site and data monitoring, including the usage of health electronic records to keep the trials moving forward at the patient's convenience while maintaining the patient safety.
Use relevant channels of communication to spread awareness among participants with diverse backgrounds: Overcoming language barriers is crucial when conducting research with diverse communities. Initiatives such as introducing comprehensive online resources for patients, researchers, and clinical trial sponsors to access publications, datasets, news, and links to the active global clinical trials, can help participants get more clarity about the topic and better understand the process.
Re-align internal processes and business strategies with diversity as a core focus: Industry stakeholders can address the diversity gap by re-evaluating their business strategies and re-aligning them with diversity as a focus. Re-alignment also includes restructuring budget allocations and training researchers and investigators to be fully equipped to adapt to a new style of working.
Efforts to increase engagement between patients and other stakeholders including the community, government, academicians, and pharmaceutical industry partners can help bridge the gap of representation and inclusivity in clinical trials. Fostering meaningful partnerships to build trust, co-creating strategies, and solutions, and promoting change for collective impact can revolutionize the future of the CRO industry.
Disclaimer: The views expressed in the above article are solely those of the author/agency in his/her private capacity and DO NOT represent the views of Medical Dialogues.
Read website full disclaimer here
Sanjay Vyas is the Executive Vice President and Managing Director of Parexel India. Sanjay holds an MBA with a Master’s Degree in Marketing Management & a Bachelor’s Degree in Commerce & Economics from the University of Mumbai, India.


