- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Cuba in third stage trial process for advanced prostate cancer vaccine
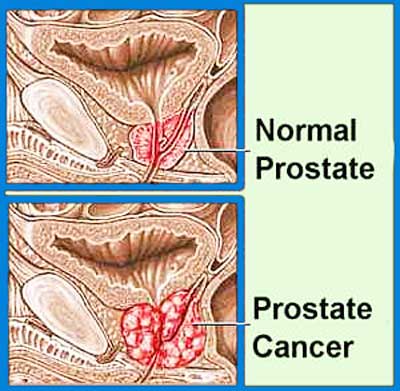
A Cuban vaccine, currently under trial stage process will now enter its third phase of the trail process, to be tested on 300 patients, confirmed the specialists to PTI. The vaccine's first phase of trial took place in 2007 and the second phase concluded in early 2015.
In this stage, researchers at the Centre for Genetic Engineering and Biotechnology (CIGB), where the vaccine Heberprovac was developed, will compare its performance with that of Zoladex, a well known therapy used internationally to treat various types of neoplasms, reported Xinhua.
Researchers from CIBG consider Heberprovac to be already superior to Zoladex in its administration format, needing only seven shots; while the Zoladex, which is also used to treat breast cancer, must be administered every three months via a sub-cutaneous implant.
The specialists say that while Heberprovac has not yet been proven to be a cure for prostate cancer, it has led to "a greater survival rate and better quality of life" in the majority of patients treated with it.
In this stage, researchers at the Centre for Genetic Engineering and Biotechnology (CIGB), where the vaccine Heberprovac was developed, will compare its performance with that of Zoladex, a well known therapy used internationally to treat various types of neoplasms, reported Xinhua.
Researchers from CIBG consider Heberprovac to be already superior to Zoladex in its administration format, needing only seven shots; while the Zoladex, which is also used to treat breast cancer, must be administered every three months via a sub-cutaneous implant.
The specialists say that while Heberprovac has not yet been proven to be a cure for prostate cancer, it has led to "a greater survival rate and better quality of life" in the majority of patients treated with it.
Next Story


