- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Guselkumab improves quality of life in patients with moderate-to-severe psoriasis, Study says
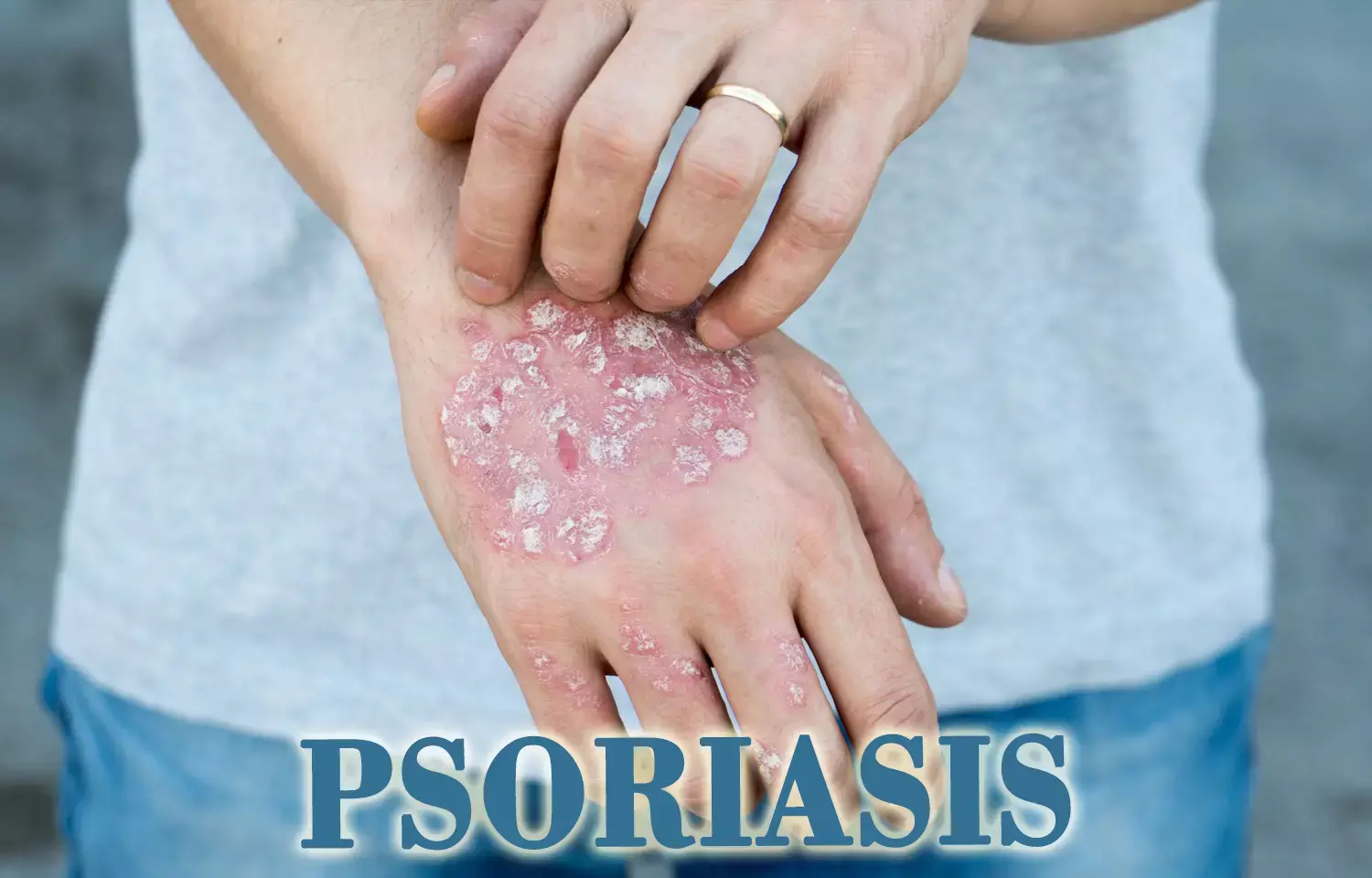
According to recent research, it has been found out that guselkumab maintains high levels of clinical response and patient-reported outcome (PRO) improvement through 5 years in patients with moderate-to-severe psoriasis.
The study is published in the British Journal of Dermatology.
Psoriasis is a chronic disease requiring long-term therapy. Hence, K. Reich and colleagues from the Translational Research in Inflammatory Skin Diseases, Institute for Health Services Research in Dermatology and Nursing, University Medical Center Hamburg-Eppendorf, Germany carried out the present study with the objective to evaluate physician- and patient-reported outcomes (PROs) through week 252 in VOYAGE 1 and VOYAGE 2.
A total of 1829 patients were randomized at baseline to receive guselkumab 100 mg every-8-weeks, placebo, or adalimumab; placebo patients crossed-over to guselkumab at week 16. Adalimumab patients crossed-over to guselkumab at week 52 in VOYAGE 1, and randomized-withdrawal/retreatment occurred at weeks 28-76 in VOYAGE 2; all patients then received open-label guselkumab through week 252.
Efficacy and HRQoL endpoints were analyzed through week 252. Safety was monitored through week 264.
The following results were observed-
a. The proportions of patients in the guselkumab group who achieved clinical responses at week 252 in VOYAGE 1 and VOYAGE 2, respectively, were: 84.1% and 82.0% (PASI90); 82.4% and 85.0% (IGA0/1); 52.7% and 53.0% (PASI100); and 54.7% and 55.5% (IGA0).
b. HRQoL endpoints were achieved as follows: 72.7% and 71.1% of patients (Dermatology Life Quality Index 0/1 or no effect on patient's life); 42.4% and 42.0% (Psoriasis Symptoms and Signs Diary [PSSD] symptom score=0); and 33.0% and 31.0% (PSSD sign score=0).
c. In VOYAGE 2 only, approximately 45% of patients achieved ≥5-point reduction in SF-36 Physical/Mental component scores and 80% reported no anxiety/depression (Hospital Anxiety and Depression scores <8).
d. Similar findings were reported for adalimumab crossovers.
e. These effects were maintained from week 52 in VOYAGE 1 and week 100 in VOYAGE 2.
f. No new safety signals were identified.
Therefore, the authors concluded that "Guselkumab maintains high levels of clinical response and PRO improvement through 5 years in patients with moderate-to-severe psoriasis."
Dr. Nandita Mohan is a practicing pediatric dentist with more than 5 years of clinical work experience. Along with this, she is equally interested in keeping herself up to date about the latest developments in the field of medicine and dentistry which is the driving force for her to be in association with Medical Dialogues. She also has her name attached with many publications; both national and international. She has pursued her BDS from Rajiv Gandhi University of Health Sciences, Bangalore and later went to enter her dream specialty (MDS) in the Department of Pedodontics and Preventive Dentistry from Pt. B.D. Sharma University of Health Sciences. Through all the years of experience, her core interest in learning something new has never stopped. She can be contacted at editorial@medicaldialogues.in. Contact no. 011-43720751
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


