- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
New treatment for Molluscum Contagiosum receives FDA approval
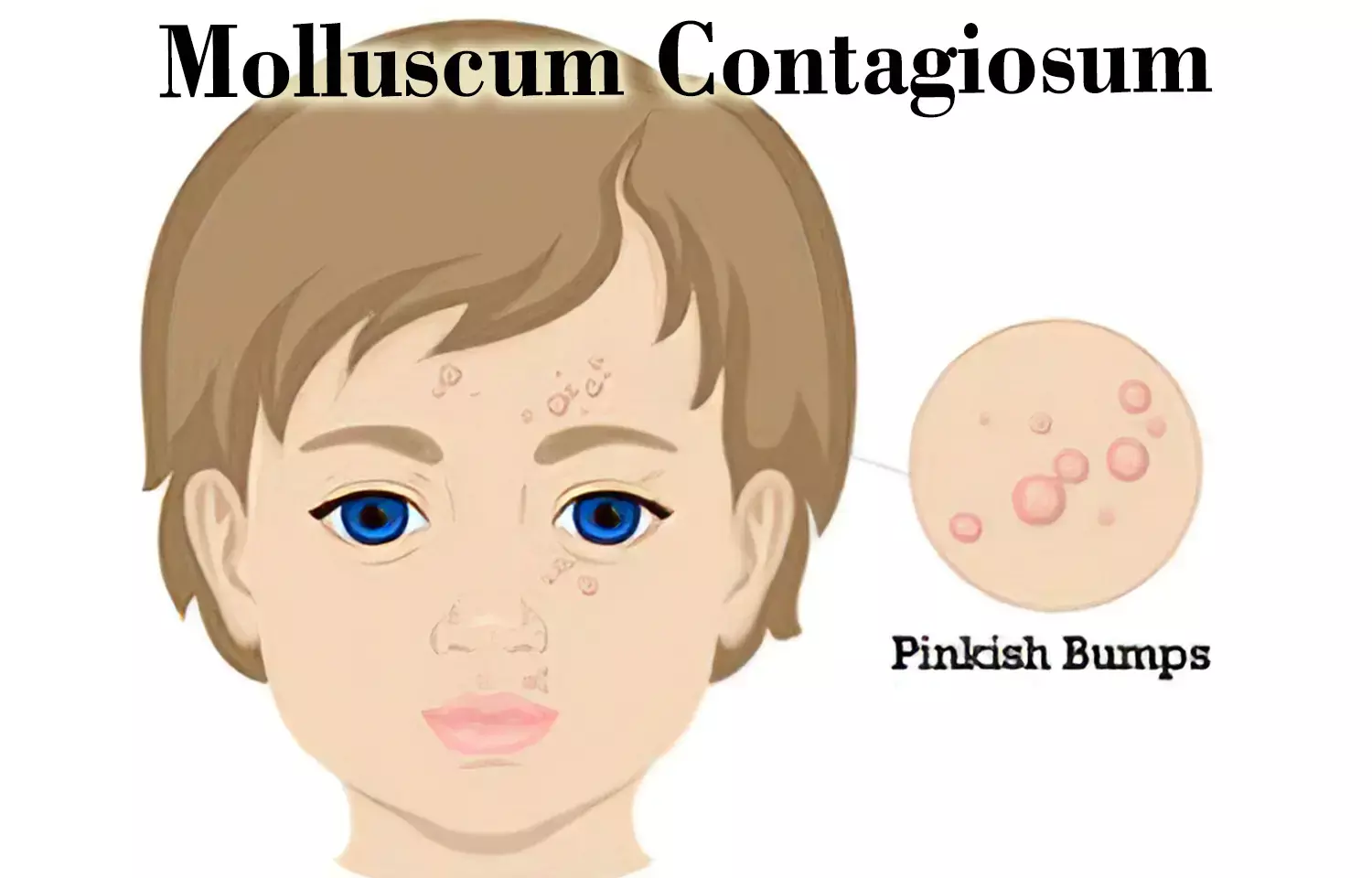
USA: VP-102 (YCANTHE), the first treatment indicated for viral-borne skin disease molluscum contagiosum, has received approval from the US Food and Drug Administration (FDA).
The drug-device combination product is designed through a single-use applicator for topical dosing and targeted delivery. It was developed with a controlled formulation of cantharidin (0.7% w/v).
“We are proud to bring patients and caregivers the first FDA-approved treatment for molluscum, which is one of the largest unmet needs in medical dermatology,” Ted White, Verrica president and chief executive officer, said in the accompanying statement. “Verrica is the first company to develop a proprietary applicator and GMP formulation of cantharidin that allows a safe, effective and precise topical administration, and the first company to successfully gain FDA approval after conducting rigorous clinical trials to evaluate the safe and effective use of a cantharidin-based product for the treatment of molluscum."
The New Drug Application (NDA) for this new treatment had been backed by data from the CAMP-1 and CAMP-2 clinical trials, which had shown a substantially higher rate of complete clearance of molluscum lesions and reduction in lesion count compared to the control arm at 84 days (P <.0001).2
Additional post-hoc analyses of the CAMP trials showed that a statistically significant greater proportion of the VP-102 arm achieved complete lesion clearance than vehicle across all affected body regions, including areas deemed most sensitive.
During these 2 trials, certain adverse events were noted in some of the participants who had been given VP-102, including pruritus, blistering, pain, and erythema at the application site. No serious adverse events were reported by treated patients.3
The PDUFA date for the drug’s approval had been set in February of 2023 when Verrica Pharmaceuticals accepted the NDA for VP-102.
Verrica had initially submitted their application for VP-102 to the FDA for this specific indication in 2021, but they received a Complete Response Letter (CRL) as a result of observed deficiencies at a contract manufacturing organization facility that was not involved in the drug's production.
However, the CRL announcement by Verrica emphasized that the treatment’s safety, clinical, or manufacturing properties were not found to be deficient by the FDA at the time. Besides its current use for this indication, VP-102 is also being assessed in another 2 mid-stage clinical trials for the treatment of common warts and external genital warts.
Recent late-breaking phase 3 data from B-SIMPLE showed berdazimer gel 10.3% treatment led to about 75% improvement in complete lesion clearance compared to vehicle and, specifically among children aged 6 to <12 years old, a complete clearance rate of 31.1% complete clearance rate versus 14.0% at 12 weeks.4
These findings, presented at the American Academy of Dermatology (AAD) 2023 Annual Meeting in New Orleans, demonstrate that VP-102 and topical berdazimer are the 2 most promising potential treatments for molluscum contagiosum among children and adults.
Molluscum contagiosum is a pox virus-borne infection known to affect approximately 6 million people in the US, with children being the most commonly affected population and the greatest incidence being in children 1-14 years old.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


