- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
GLP1RAs associated with reduced amputation rate and CV risk in diabetics: Study
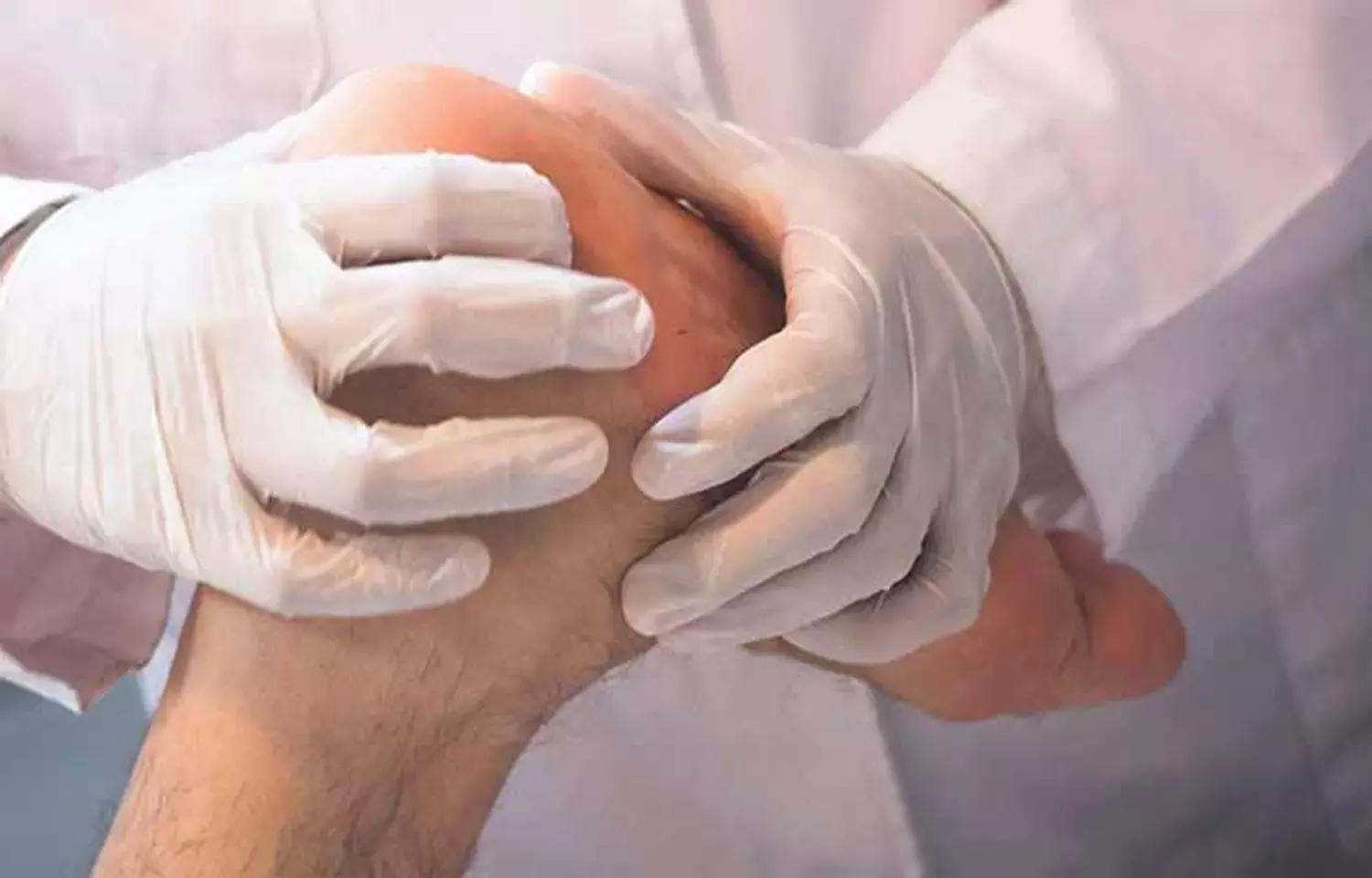
Treatment with GLP1RAs, when compared with DPP4is, was associated with significantly lower composite adverse cardiovascular outcomes and major adverse limb events among individuals with type 2 diabetes.
Taiwan: The use of Glucagon-like peptide-1 receptor agonists (GLP1RAs) is associated with a significantly lower risk of major adverse limb events in patients with type 2 diabetes compared to dipeptidyl peptidase-4 inhibitors (DPP4is), a recent study has found. The risk reduction was largely driven by a reduced rate of amputations.
"Also, treatment with GLP1RAs was associated with a lower risk of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke, and death from any cause, the researchers wrote in the journal Diabetologia.
Glucagon-like peptide-1 receptor agonists (GLP1RAs) and dipeptidyl peptidase-4 inhibitors (DPP4is) are a group of medications used for the treatment of type 2 diabetes. Previous cardiovascular outcome trials have established the safety and efficacy of GLP1RAs and DPP4is in major cardiovascular adverse events. However, the effects of these drugs on adverse limb outcomes are poorly examined.
Considering the above, Jen-Kuang Lee, Cardiovascular Center, National Taiwan University Hospital, Taipei, Taiwan, and colleagues aimed to determine the real-world outcomes of patients with diabetes mellitus receiving GLP1RAs as compared with those receiving DPP4is in terms of major adverse cardiovascular and limb events.
For this purpose, the researchers conducted a retrospective cohort study with data collected by the Taiwan National Health Insurance database between 1 May 2011 and 31 December 2017. During this period, patients who were treated for type 2 diabetes with a GLP1RA or DDP4i were identified (n = 1,080,993).
The primary outcome was a composite of major adverse limb events, defined as peripheral artery disease (PAD), critical limb ischemia, percutaneous transluminal angioplasty or peripheral bypass for PAD, and amputation.
A total of 948,342 people were identified, 4460 in the GLP1RA group and 13,380 in the DPP4i group after propensity-score matching.
The study yielded the following findings:
- The incidence of primary composite outcome events was significantly lower in those treated with GLP1RAs compared with those treated with DPP4is (2.59 vs 4.22 events per 1000 person-years; subdistribution HR [SHR] 0.63), primarily due to lower rates of amputation (1.29 events per 1000 person-years for GLP1RAs vs 2.4 events per 1000 person-years for DPP4is; SHR 0.55).
- Treatment with GLP1RAs was also associated with significantly lower risks of secondary composite outcome events (11.02 vs 17.95 events per 1000 person-years; HR 0.62).
- The observed beneficial effects of GLP1RAs on reducing composite adverse limb outcomes were particularly noticeable in the non-cardiovascular patients and statin users.
The researchers concluded, "some unexplored confounding factors may exist in this observation study and future large-scale randomised controlled trials are needed."
Reference:
The study titled, "Major adverse cardiovascular and limb events in patients with diabetes treated with GLP-1 receptor agonists vs DPP-4 inhibitors," is published in the journal Diabetologia.
DOI: https://link.springer.com/article/10.1007/s00125-021-05497-1
MSc. Biotechnology
Medha Baranwal holds a Bachelor’s degree in Biomedical Sciences from the University of Delhi and a Master’s degree in Biotechnology from Amity University. Since May 2018, she has been contributing to Medical Dialogues, writing and editing medical news articles that translate complex research into clear, accessible information for healthcare professionals.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


