- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Is Using Proton Pump Inhibitors Depriving Us of Essential Micronutrients?
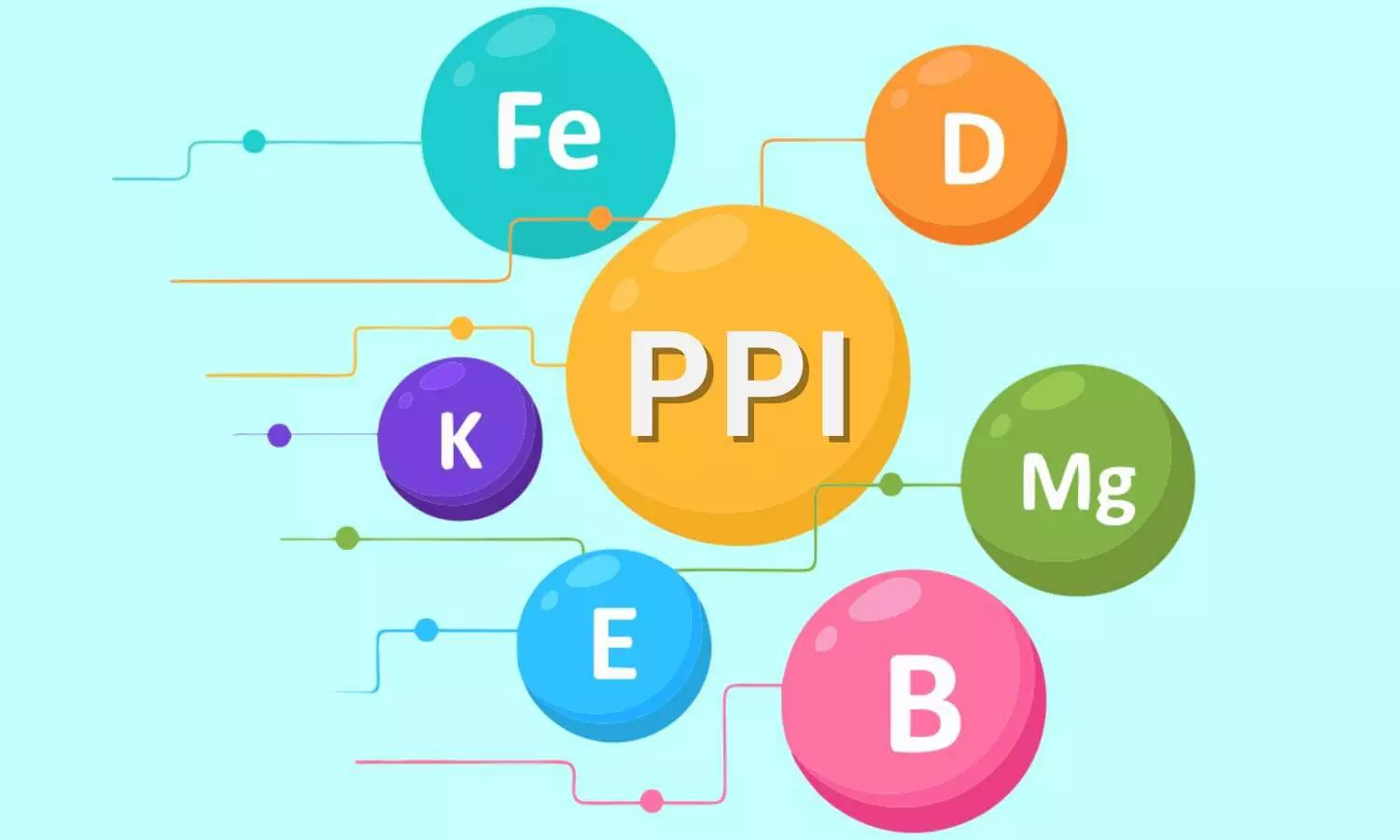
The ubiquitous presence and widespread utility of proton pump inhibitors (PPIs) among general physicians and gastroenterologists are well established.[1] These class of drugs work by reducing acid production in the stomach, thus healing peptic ulcers, gastroesophageal reflux disease (GERD), Barrett's esophagus, Zollinger-Ellison syndrome, and the eradication of Helicobacter pylori as part of combination regimens.[2]
Although PPIs continue to be universally heralded as an effective pharmacotherapeutic option and are preferred by many practicing physicians, they come with certain unfavorable adverse effects when used for long-term. The US Food and Drug Administration (US-FDA) issued a warning in 2011 postulating a relationship between long-term PPI use and micronutrient deficiency (hypomagnesemia).[3]
This article aims to give an in-depth perspective on how PPIs, in the long run, can be a potential risk factor leading to micronutrient deficiency.
Potential Mechanism of PPIs and Micronutrient Deficiency in the Long Run[3,4,5]
PPIs are among the most widely used medications worldwide, having high prescription rates, especially among the elderly. The mechanism of PPI-induced micronutrient deficiency is likely multifactorial and includes reduced stomach acid secretion and changes in the gut microbiome.
Stomach acid is vital in absorbing several micronutrients, including iron, calcium, magnesium, and vitamin B12. PPIs' mode of action is based on suppressing the H+/K+ ATPase enzyme in gastric mucosal parietal cells, altering the bioavailability and absorption of critical vitamins and minerals in both the stomach and duodenum, thereby affecting their distal absorption.
The prolonged use of PPIs can cause changes in the gut microbiome, leading to bacterial overgrowth or changes in bacterial species. These changes can also impact the absorption of certain nutrients, as some bacteria play a role in synthesizing or breaking down certain micronutrients.
PPIs have been linked to an increased risk of vitamin and mineral deficiencies affecting vitamin B12, vitamin C, calcium, iron, and magnesium metabolism. These concerns are more noticeable in elderly and malnourished patients and those on chronic hemodialysis and concomitant PPI therapy.
Clinical Evidence Supporting Causality of PPIs Use and Micronutrient Deficiency
Several studies have suggested that long-term use of proton pump inhibitors (PPIs) may be associated with micronutrient deficiencies.
Magnesium: Magnesium is an essential intracellular cation that plays a role in multiple cellular activities. All PPIs that are biochemically substituted pyridyl methyl sulphonyl benzimidazole derivatives (in order of potency: rabeprazole, esomeprazole, omeprazole, lansoprazole, and pantoprazole) have been linked to hypomagnesemia. [3]
- A meta-analysis conducted by Srinutta T. et al. noted the association of PPI use and development of hypomagnesemia. The meta-analysis included 16 observational studies, including 13 cross-sectional studies, two case-control studies, and one cohort study, adding a total of 131,507 patients. The pooled percentage of PPI users was 43.6% (95% confidence interval [CI] 25.0%, 64.0%). Among PPI users, 19.4% (95% CI 13.8%, 26.5%) had hypomagnesemia compared to 13.5% (95% CI 7.9%, 22.2%) among nonusers. In subgroup analyses, high-dose PPI use was associated with higher odds for hypomagnesemia than low-dose PPI use (pooled adjusted OR 2.13; 95% CI 1.26, 3.59; P = .005), hence stressing the causality of PPIs and hypomagnesemia. [6]
- Another cross-sectional study by Gau et al. also revealed the potential effects of PPI therapy relative to the development of hypomagnesemia. The study included 207 hospitalized adults on PPI therapy aged over 50 years and their serum magnesium were measured. The study concluded that standard and high-dose PPI use was associated with an increased risk of hypomagnesemia (adjusted OR = 2.50, 95% CI = 1.43–4.36), suggesting a subclinical insufficiency or deficiency status in asymptomatic individuals. [7]
Vitamin B12: Vitamin B12 (Cobalamin) is a water-soluble essential nutrient. Peptic enzymes break dietary B12 from dietary proteins during vitamin B12 absorption. In the absence of gastric acid, vitamin B12 cannot be cleaved from food protein and hence cannot bind to R-proteins, which protects vitamin B12 from pancreatic digestion. [3]
- An observational study carried out by Porter KM et al noted the higher risk of Vitamin B12 deficiency among PPI users. The study involving a sample size of 3299 subjects, of which 1216 were PPI users and 2083 non-PPI users. The primary outcome was serum total vitamin B12, homocysteine and methylmalonic acid (MMA), and holotranscobalamin (holo-TC) concentration levels. The study concluded 25% of PPI users have a high prevalence of vitamin B12 deficiency compared to 15% of controls (p<0.001). Also, high PPI doses (≥30mg/d) were associated with low holoTC and high MMA concentration (p<0.001), suggesting PPI use was associated with an increased risk of vitamin B12 deficiency. [8]
- Lewis JR et al.in their prospective cohort also highlighted the lower total serum vitamin B12 levels among PPI users. The study enrolled 94 subjects, of which 46 were long-term PPI users, and 48 were controls (non-PPI users). The primary outcome measured was serum B12 levels. The study concluded that twenty-three of 46 (50%) PPI users had low total serum vitamin B12 levels compared to 10 of 48 (20.8%) non-PPI users, p= 0.003; PPI users had 18% lower total serum vitamin B12 levels compared to controls. (p=0.030). [9]
Vitamin C: Since vitamin C is an essential nutrient, appropriate amounts of water-soluble vitamins must be obtained through dietary consumption. Administration of PPIs reduces its bioavailability by decreasing the concentration of vitamin C in gastric secretions and the fraction of vitamin C in its active antioxidant form. [3,10]
- Study indicated that the association of vitamin C deficiency and use of PPI. Mowat et al. conducted their study on 20 healthy volunteers (9 positive for Helicobacter pylori) who were analyzed for ascorbic acid and total vitamin C before and were repeated after four weeks of treatment with omeprazole, 40 mg daily. The study concluded that omeprazole decreased fasting gastric ascorbic acid levels from 3.8 to 0.7 microg/mL (P < 0.001). In H. pylori-infected subjects, omeprazole also decreased total vitamin C levels in both gastric juice and serum, suggesting administration of PPIs can cause vitamin C deficiency. [10]
Calcium: It has been postulated that stomach acid and the proximal duodenum's slightly acidic milieu are required to dissociate ingested calcium from a meal bolus, allowing it to be absorbed. Without this milieu, elemental calcium is not absorbed, which may result in compensatory physiologic alterations such as secondary hyperparathyroidism.[3] PPIs also inhibit the vacuolar-type of H+-ATPase of osteoclasts. This process might cause aberrant osteoclast-mediated bone resorption and osteoporosis.[11]
- O'Connell et al. in their randomized study found that 20 mg of omeprazole reduced fractional calcium absorption by 9.1% (95% CI: 6.5-11.6%) compared to 3.5% (95% CI: 1.6-5.5%) induced by placebo in a randomized, double-blind, placebo-controlled, cross-over experiment involving 18 women (mean age of 76 ± 7 years) suggesting PPIs can hamper calcium absorption from the gut.[12]
- Analytical cross-sectional study by Fattahi, M. R. et al. found out the lower level of bone mineral content (BMC) among PPI users. The study recruited 394 participants in which 133 subjects used PPIs for more than two years, and the control group was non-PPI users (n=261). The median duration of PPI use was 6.7 (2–31) years. There were significant differences in the T-scores (used to compare the bone density of an individual with that of an average young person with healthy bones) between the two groups for the femoral neck (P<0.001). Osteoporosis in the femoral neck was significantly higher in PPI users than in the comparator group suggesting PPIs can interfere with calcium absorption from the gut. The study concluded that BMC levels were significantly lower in PPI users in all three regions: lumbar spine, total hip, and femoral neck (P<0.001).[13]
Iron: Dietary iron is provided in meals as either nonheme (66%) or heme iron (32%), and stomach acid significantly improves nonheme iron absorption. It has been postulated that chronic PPI medication causes clinically significant iron malabsorption due to stomach acid hyposecretion and the risk of achlorhydria.[3]
- A retrospective cohort study carried out by Sarzynski E. et al highlighted the association between chronic PPI use (who received the therapy for at least one year) and iron malabsorption. The study included 98 patients, and PPI users were observed to have significant decrease in mean hemoglobin and hematocrit concentrations (p < 0.01 for both parameters) compared with matched controls. The odds ratio of decreasing hemoglobin by 1.0 g/dl while on chronic PPI therapy was 5.03 (95% CI = 1.71–14.78, p < 0.01), while the odds ratio of decreasing hematocrit by 3% was 5.46 (95% CI = 1.67–17.85, p < 0.01) suggesting the potent role of PPIs in iron malabsorption. [14]
Conclusion
Although PPIs have revolutionized the therapy of numerous upper gastrointestinal tract disorders, their use brings along their caveats; long-term use of PPIs has been associated with micronutrient deficiencies. There is compelling evidence substantiating this concern. Risk stratification of elderly, frail, malnourished, dialyzed, and chronically hospitalized patients could guide physicians to weigh the benefits of therapy versus its risks. Hence, clinicians must continuously assess their patients' needs to continue long-term PPI therapy and timely deprescribe their PPI prescription with on demand use of H2RAs.
References
1. Strand, D. S., Kim, D., & Peura, D. A. (2017). 25 Years of Proton Pump Inhibitors: A Comprehensive Review. Gut and Liver, 11(1), 27-37.
2. Shin, J. M., & Sachs, G. (2008). Pharmacology of Proton Pump Inhibitors. Current gastroenterology reports, 10(6), 528.
3. Heidelbaugh, J. J. (2013). Proton pump inhibitors and risk of vitamin and mineral deficiency: Evidence and clinical implications. Therapeutic Advances in Drug Safety, 4(3), 125-133.
4. Vinke P, Wesselink E, van Orten-Luiten W, van Norren K. The Use of Proton Pump Inhibitors May Increase Symptoms of Muscle Function Loss in Patients with Chronic Illnesses. International Journal of Molecular Sciences. 2020; 21(1):323.
5. Freedberg DE, Lebwohl B, Abrams JA. The impact of proton pump inhibitors on the human gastrointestinal microbiome. Clin Lab Med. 2014 Dec;34(4):771-85.
6. Srinutta, T., Chewcharat, A., Takkavatakarn, K., Praditpornsilpa, K., Eiam-Ong, S., Jaber, B. L., & Susantitaphong, P. (2019). Proton pump inhibitors and hypomagnesemia: A meta-analysis of observational studies. Medicine, 98(44), e17788.
7. Gau JT, Yang YX, Chen R, Kao TC. Uses of proton pump inhibitors and hypomagnesemia. Pharmacoepidemiol Drug Saf. 2012;21(5):553-559.
8. Lewis JR, Barre D, Zhu K, et al. Long-term proton pump inhibitor therapy and falls and fractures in elderly women: a prospective cohort study. J Bone Miner Res. 2014;29(11):2489-2497.
9. Porter KM, Hoey L, Hughes CF, et al. Associations of atrophic gastritis and proton-pump inhibitor drug use with vitamin B-12 status, and the impact of fortified foods, in older adults. Am J Clin Nutr. 2021;114(4):1286-1294.
10. Mowat, C., Carswell, A., Wirz, A., & McColl, K. E. (1999). Omeprazole and dietary nitrate independently affect levels of vitamin C and nitrite in gastric juice. Gastroenterology, 116(4), 813–822.
11. Jo Y, Park E, Ahn SB, Jo YK, Son B, Kim SH, Park YS, Kim HJ. A Proton Pump Inhibitor's Effect on Bone Metabolism Mediated by Osteoclast Action in Old Age: A Prospective Randomized Study. Gut Liver. 2015 Sep 23;9(5):607-14.
12. Thong, B. K. S., Ima-Nirwana, S., & Chin, K. Y. (2019). Proton Pump Inhibitors and Fracture Risk: A Review of Current Evidence and Mechanisms Involved. International journal of environmental research and public health, 16(9), 1571.
13. Fattahi, M. R., Niknam, R., Shams, M., Anushiravani, A., Taghavi, S. A., Omrani, G. R., & Mahmoudi, L. (2019). The Association Between Prolonged Proton Pump Inhibitors Use and Bone Mineral Density. Risk management and healthcare policy, 12, 349–355.
14. Sarzynski E, Puttarajappa C, Xie Y, Grover M, Laird-Fick H. Association between proton pump inhibitor use and anemia: a retrospective cohort study. Dig Dis Sci. 2011;56(8):2349-2353.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


