- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
COBRRA Trial: Apixaban Shows Lower Bleeding Risk Than Rivaroxaban in Acute VTE
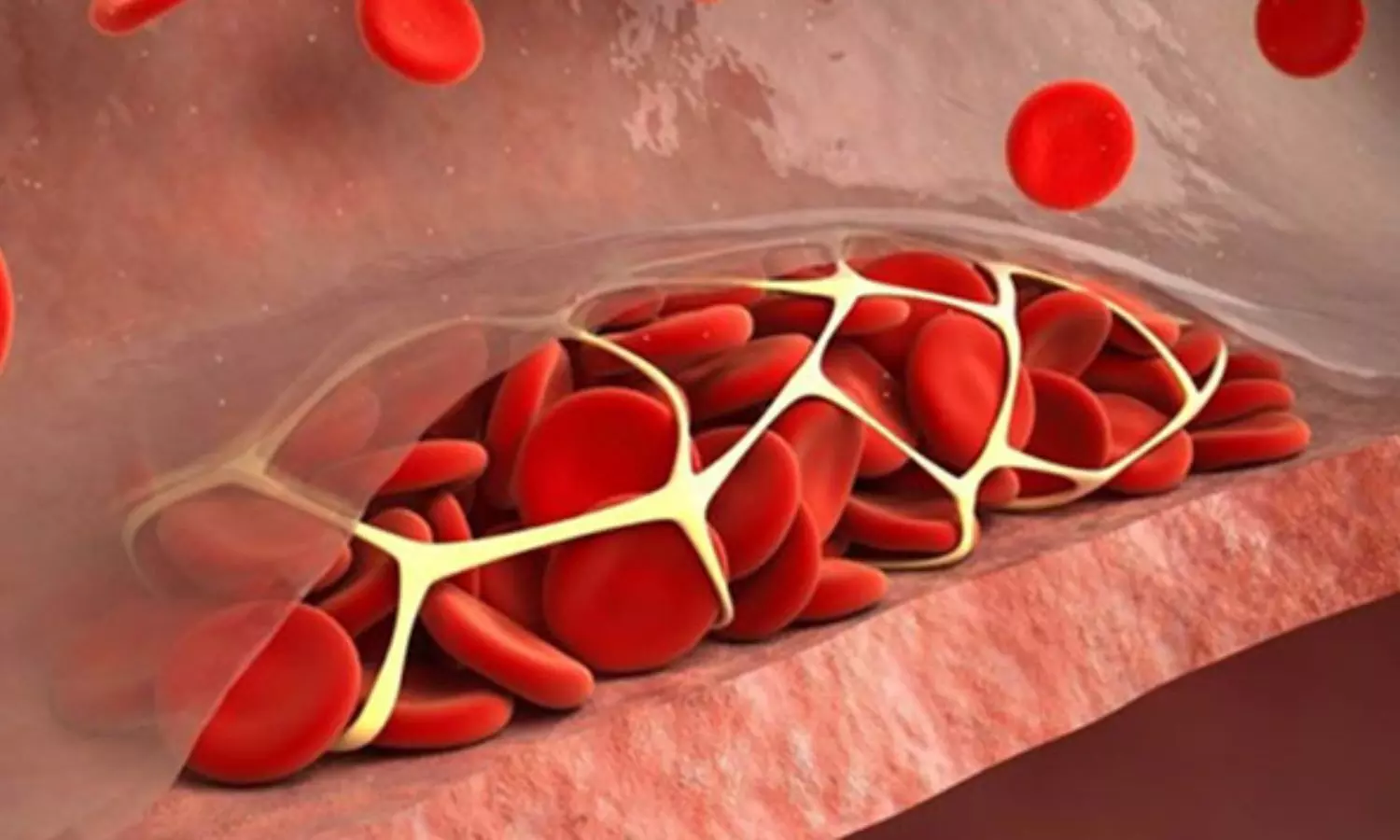
The COBRRA randomized trial that compared direct oral anticoagulants (DOACs) for acute venous thromboembolism (VTE), and found apixaban to be associated with a significantly lower risk of clinically relevant bleeding than rivaroxaban during the 3-month treatment period. The findings were published in The New England Journal of Medicine.
Both medications belong to a class of drugs known as DOACs and are widely prescribed globally against blood clots. However, physicians have long debated whether one carries a higher bleeding risk than the other, since anticoagulants can increase the risk of bleeding complications, while preventing clots.
Thus, this randomized clinical trial involving 2,760 patients diagnosed with acute symptomatic pulmonary embolism or proximal deep-vein thrombosis was carried out. The participants were randomly assigned to receive either apixaban or rivaroxaban for a treatment period of 3 months.
The trial had blinded evaluation of outcomes in patients assigned to apixaban received 10 mg twice daily for the first 7 days, followed by 5 mg twice daily. Those in the rivaroxaban group received 15 mg twice daily for 21 days, followed by 20 mg once daily. The primary goal of this study was clinically relevant bleeding during the 3-month treatment period. This included both major bleeding and clinically relevant non-major bleeding as defined by the International Society on Thrombosis and Haemostasis.
Clinically relevant bleeding occurred in 44 (3.3%) of 1,345 patients in the apixaban group and, 96 (7.1%) of 1,355 patients receiving rivaroxaban. The relative risk of this was translated to 0.46 and the bleeding risk in apixaban group was roughly half when compared to the rivaroxaban group.
All-cause mortality occurred in one patient in the apixaban group (0.1%) and 4 patients in the rivaroxaban group (0.3%). Although the numbers were small and the difference was not statistically conclusive, the results indicated no apparent safety disadvantage for apixaban.
Serious adverse events unrelated to bleeding or clot recurrence were reported in 36 patients (2.7%) receiving apixaban and 30 patients (2.2%) receiving rivaroxaban, which suggested broadly comparable safety profiles aside from bleeding risk.
While both medications remain effective options for preventing clot progression and recurrence, the lower bleeding risk observed with apixaban may influence prescribing decisions, particularly for patients already considered at higher risk of bleeding complications.
Reference:
Castellucci, L. A., Chen, V. M., Kovacs, M. J., Lazo-Langner, A., Greenstreet, P., Kahn, S., Côté, B., Schulman, S., de Wit, K., Douketis, J., Suryanarayan, D., Wan, T., Yeo, E., Le Templier, G., Tran, H. A., Willcox, A., Crowther, H. J., Prasad, R., Shivakumar, S., … Rodger, M. (2026). Bleeding risk with apixaban vs. Rivaroxaban in acute venous thromboembolism. The New England Journal of Medicine, 394(11), 1051–1060. https://doi.org/10.1056/nejmoa2510703
Neuroscience Masters graduate
Jacinthlyn Sylvia, a Neuroscience Master's graduate from Chennai has worked extensively in deciphering the neurobiology of cognition and motor control in aging. She also has spread-out exposure to Neurosurgery from her Bachelor’s. She is currently involved in active Neuro-Oncology research. She is an upcoming neuroscientist with a fiery passion for writing. Her news cover at Medical Dialogues feature recent discoveries and updates from the healthcare and biomedical research fields. She can be reached at editorial@medicaldialogues.in
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


