- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Saliva swab as good as nasal swab for diagnosing COVID-19 infection, finds NEJM study
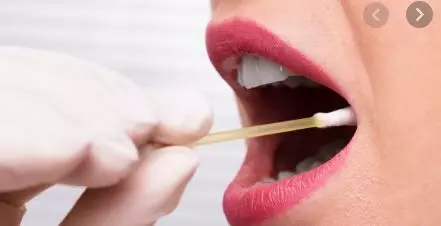
Saliva swabs have shown equal efficacy to nasopharyngeal swabs in tests to confirm infection with SARS-CoV-2, suggests a recent study.
The findings of the study have been published in the New England Journal of Medicine.
Rapid and accurate diagnostic tests are essential for controlling the ongoing Covid-19 pandemic. Although the current standard involves testing of nasopharyngeal swab specimens by quantitative reverse-transcriptase polymerase chain reaction (RT-qPCR) to detect SARS-CoV-2, saliva specimens may be an alternative diagnostic sample.
To determine how saliva specimens compare with nasopharyngeal swab specimens concerning sensitivity in the detection of SARS-CoV-2 during infection, a team of researchers under Anne L. Wyllie, at the Yale School of Public Health, New Haven, sought to carry out a study with 70 COVID 19 positive patients.
Between March 23rd and June 16th, 2020, 321 COVID-19 inpatients at Yale-New Haven Hospital were identified as being eligible to enroll in our study and were approached by the clinical team. Of these, 202 individuals were enrolled. Also, 495 asymptomatic healthcare workers (aged 22-74 years, average = 37.6 years; male, n = 105, 21%) were also enrolled in the active monitoring protocol.
The collection of nasopharyngeal and saliva samples was attempted every three days throughout their clinical course. Saliva samples were self-collected by the patient.
Results revealed the following facts.
- More SARS-CoV-2 RNA copies were detected in the saliva specimens (mean log copies per milliliter, 5.58; 95% confidence interval [CI], 5.09 to 6.07) than in the nasopharyngeal swab specimens (mean log copies per milliliter, 4.93; 95% CI, 4.53 to 5.33).
- Also, a higher percentage of saliva samples than nasopharyngeal swab samples were positive up to 10 days after the Covid-19 diagnosis.
- At 1 to 5 days after diagnosis, 81% (95% CI, 71 to 96) of the saliva samples were positive, as compared with 71% (95% CI, 67 to 94) of the nasopharyngeal swab specimens.
- The level of SARS-CoV-2 RNA decreased after symptom onset in both saliva specimens (estimated slope, −0.11; 95% credible interval, −0.15 to −0.06) and nasopharyngeal swab specimens (estimated slope, −0.09; 95% credible interval, −0.13 to −0.05).
- During the clinical course, less variation was observed in levels of SARS-CoV-2 RNA in the saliva specimens (standard deviation, 0.98 virus RNA copies per milliliter; 95% credible interval, 0.08 to 1.98) than in the nasopharyngeal swab specimens (standard deviation, 2.01 virus RNA copies per milliliter; 95% credible interval, 1.29 to 2.70).
"Collection of saliva samples by patients themselves negates the need for direct interaction between health care workers and patients. This interaction is a source of major testing bottlenecks and presents a risk of nosocomial infection. The collection of saliva samples by patients themselves also alleviates demands for supplies of swabs and personal protective equipment. Given the growing need for testing, our findings provide support for the potential of saliva specimens in the diagnosis of SARS-CoV-2 infection." concluded the team.
Primary source: The New England Journal of Medicine
Dr Satabdi Saha (BDS, MDS) is a practicing pediatric dentist with a keen interest in new medical researches and updates. She has completed her BDS from North Bengal Dental College ,Darjeeling. Then she went on to secure an ALL INDIA NEET PG rank and completed her MDS from the first dental college in the country – Dr R. Ahmed Dental College and Hospital. She is currently attached to The Marwari Relief Society Hospital as a consultant along with private practice of 2 years. She has published scientific papers in national and international journals. Her strong passion of sharing knowledge with the medical fraternity has motivated her to be a part of Medical Dialogues.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


