- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
NSAID use during first 22 weeks of gestation tied to increased risk of prematurity: BJOG
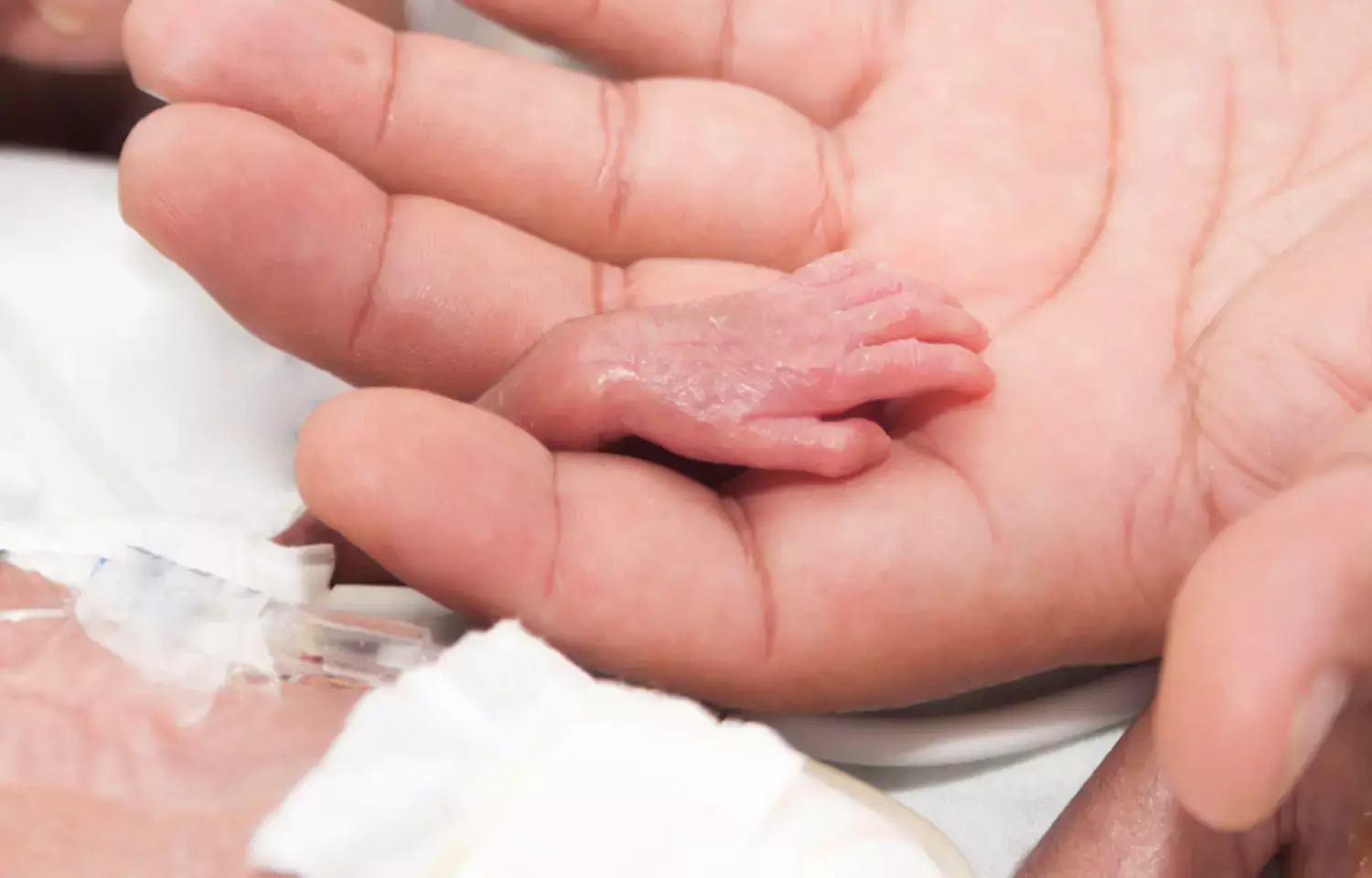
Exposure to medication during pregnancy has been shown to be frequent, particularly in France. Non-steroidal antiinflammatory drugs (NSAIDs) are widely used in the general population and are also prescribed to pregnant women who may need them to treat chronic illnesses or relieve pain (chronic or acute). The fetal consequences of late exposure to NSAIDs have been documented, particularly for premature closure of the ductus arteriosus. The global fetal toxicity of NSAIDs has led to limitation of their use during pregnancy: non-selective NSAIDs are generally contraindicated from the 6th month of pregnancy and COX-2 inhibitors throughout pregnancy leading to a significant decrease in NSAID prescription in the 3rd trimester.
However, the safety of early exposure to NSAIDs in pregnant women has not been fully established, particularly with regard to prematurity. One physio-pathological explanation associated to early exposure of pregnancy to NSAIDs could be an utero-placental hypoperfusion induced by inhibition of prostaglandin synthesis. This pathway has already been suggested as a mechanism of placental mal-implantation leading to miscarriages associated with early exposure to NSAIDs.
In this study, Quantin C and team assessed the risk of preterm birth associated with NSAIDs delivered outside of hospitals, focusing on a defined period of fixed early exposure from conception to 22 weeks of gestation. The study used the French health insurance database and included nearly 1.6 million pregnant women.
In this population-based retrospective cohort study, authors included all hospital discharge abstracts for all deliveries that occurred after 22 completed weeks of gestation in France between January 2012 and December 2014 from The French National Health Insurance.
Singleton pregnancies with a live birth occurring after 22WG from women between 15 and 45 years old and insured the year before the first day of gestation and during pregnancy were included. Authors excluded pregnancies for which anti-inflammatory medications were dispensed after 22WG. The association between exposure and risk of preterm birth was evaluated with GEE models, adjusting on a large number of covariables, socio-demographic variables, maternal comorbidities, prescription drugs and pregnancy complications.
Prematurity was defined according to the WHO classification as a birth that occurred before 37WG.
Among 15,98,330 singleton pregnancies, early exposure to non-selective NSAIDs was associated with a significantly increased risk of preterm birth, regardless of the severity of prematurity: adjusted odds ratio (aOR) = 1.76 (95% CI 1.54–2.00) for extreme prematurity (95% CI 22–27WG), 1.28 (95% CI 1.17–1.40) for moderate prematurity (28–31WG) and 1.08 (95% CI 1.05–1.11) for late prematurity (32–36WG), with non-overlapping confidence intervals.
Study identified five NSAIDs for which the risk of premature birth was significantly increased: ketoprofen, flurbiprofen, nabumetone, etodolac and indomethacin: for the latter, aOR = 1.92 (95% CI 1.37–2.70) with aOR = 9.33 (95% CI 3.75–23.22) for extreme prematurity.
In this nationwide cohort study, the frequency of prematurity was 15.5% higher following maternal exposure to at least one NSAID (including COX-2 inhibitors) delivered outside hospitals during the first 22WG, as compared with the non-exposed population. Exposure to non-selective NSAIDs in the first 22WG was associated with a significant increase in preterm births after adjustment for socio-demographic characteristics and prior conditions (aOR = 1.08).
Drug-specific analyses were made possible by the large size of the population. A significant association between the exposure to NSAIDs and preterm delivery was limited to five of the 19 NSAIDs prescribed in the first 22WG, i.e. ketoprofen, flurbiprofen, nabumetone, etodolac, and indomethacin when prescribed alone. Early exposure to selective COX-2 inhibitors (celecoxib or etoricoxib) did not increase the risk of preterm birth, nor did the two biological agents prescribed alone, etanercept and infliximab, in contrast to the non-selective NSAIDs. It is worth noting that combination of a non-selective NSAID and a biological agent was rare (42 pregnancies), but the associated risk of preterm delivery was highest in this cohort.
The overall odds ratio of 1.08 may seem weak, but study shows that the risk is much higher for extreme prematurity (1.76) and for five specific NSAIDs. The physiopathological considerations are confusing since NSAIDs are well-known tocolytic agents which direct fetal side effects in late pregnancy justify the global contraindication of NSAIDs in this period (FDA). However, some observations suggest that NSAIDs have a direct action effect on placenta circulation. Placental insufficiency, which is similar to fetal growth restriction, could have a central role in preterm delivery whatever its cause through vascular alterations (utero-placental ischaemia).
Non-selective NSAID use (delivered outside hospitals) during the first 22 weeks of pregnancy was found to be associated with an increased risk of prematurity, and particularly extreme prematurity. However, the risk of prematurity differed according to the NSAID.
This study suggests that there is an urgent need for complementary investigations, as prematurity is a frequent event, with high rates of mortality and morbidity at the lowest gestational ages, and that an assessment by INN should be preferred to assessment for the whole ATC group. These results should not be overestimated because of the risk of unjustified maternal anxiety and discontinuation of useful treatments, but neither should they be underestimated. Of course, the risk–benefit ratio between the value of these useful treatments in clinical practice and the risk of prematurity needs to be confirmed.
Additional comprehensive studies about the physiopathological consequences of prostaglandin synthesis inhibition by non-selected NSAIDs and Cox-2 inhibitors are necessary to refine the period during which these treatments may be contraindicated. The results of this study should, therefore, be considered as a pharmaco-epidemiological warning
Source: Quantin C, Yamdjieu Ngadeu C, Cottenet J, Escolano S, Bechraoui-Quantin S, Rozenberg P, Tubert-Bitter P, Gouyon J-B. BJOG 2021;128:1575–1584.
https://doi.org/10.1111/1471-0528.16732.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


