- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
The "Dark Side" of Pneumoperitoneum and Laparoscopy: A review
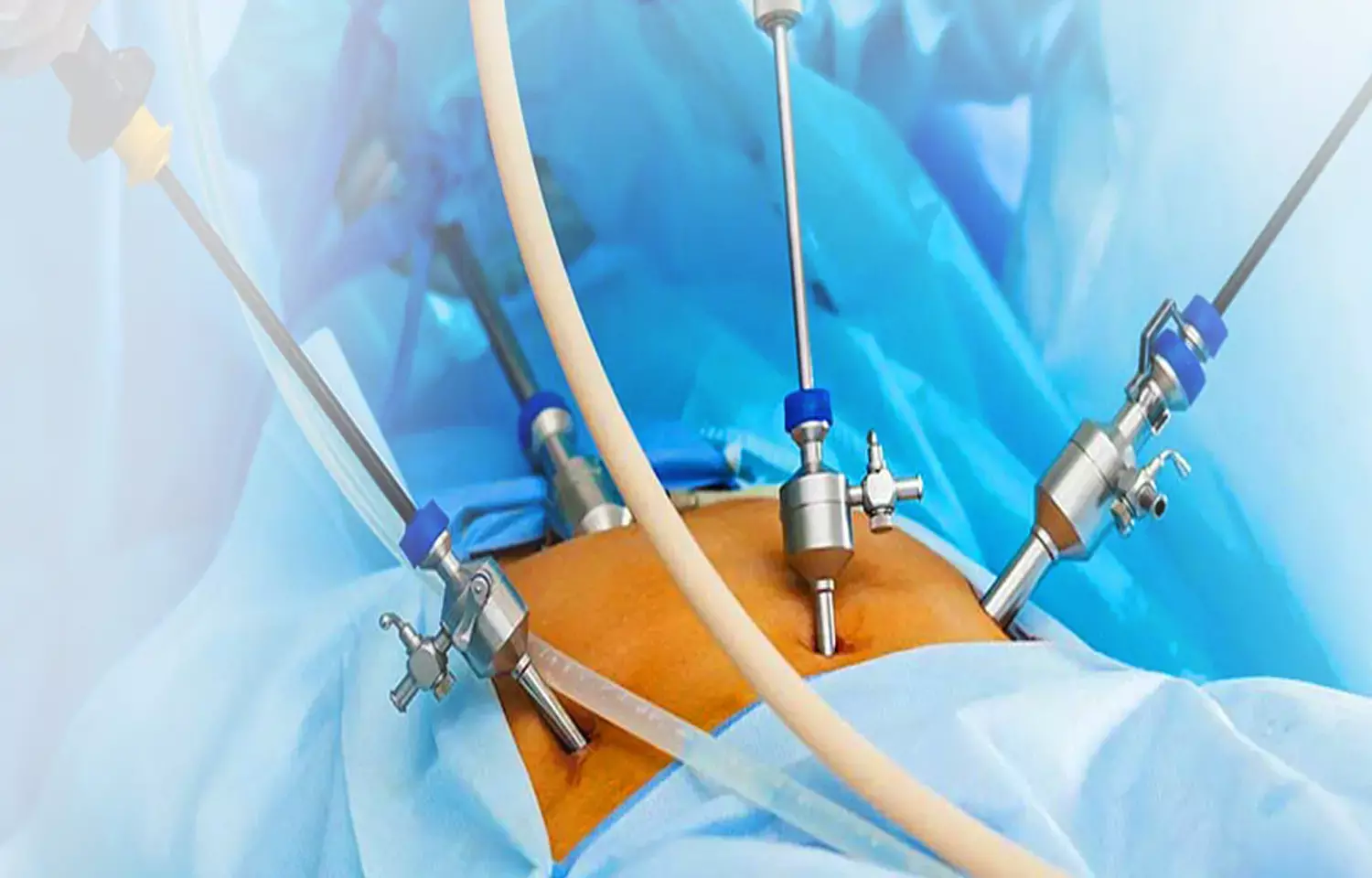
Laparoscopy has progressively substituted abdominal open surgery for many procedures as it has several advantages compared to laparotomy, even in children. In particular, laparoscopy is associated with better cosmetic results, shorter hospital stays, lower postoperative pain, and a faster return to daily activities. However, it is not free from short- and long-term comorbidities, such as light pain, abdominal discomfort, and adhesion development.
The pneumoperitoneum is essential for laparoscopic surgery, as it enables visibility and mobility in the working site. Nevertheless, the pneumoperitoneum modifies abdominal-cavity homeostasis and might promote metabolic changes through mechanical and biochemical effects.
On the basis of this evidence, CO2 has become a more suitable gas for pneumoperitoneum creation, as it is cost effective, noncombustible, and soluble, and it is rapidly excreted with respiration. However, recent studies suggest that the use of CO2 during laparoscopic surgery is not free from adverse effects. The use of this gas might induce metabolic, immune, and structural modifications that can have clinical impact.
These data suggest that chemical, physical, and biological effects of CO2 on peritoneum cause inflammation and tissue modifications. In particular, the grade of morphological changes is related to the level of intrabdominal pressure. CO2 pneumoperitoneum evokes an inflammatory response by modifying factors such as plasmatic and peritoneal chemokines levels. To further investigate these aspects of laparoscopy, Giuseppina Rosaria Umano and team reviewed the research with regards to animal and human studies investigating peritoneal and systemic alterations during and after carbon dioxide pneumoperitoneum.
They performed a narrative search of Medline and PubMed databases with terms covering "laparoscopy", "pneumoperitoneum", "inflammation", "mesothelium", "ischemia–reperfusion", and "adherence" published between January 1993 and April 2021. These terms were used in different combinations. Only English language papers were included. Articles were evaluated for scientific relevance and pertinence to the effect of pneumoperitoneum on peritoneal homeostasis impairment.
The literature search revealed 621 articles. The majority of the selected papers investigated pneumoperitoneum effects in animal models: rats (n = 21), pigs (n =4), rabbits (n = 1), and dogs (n = 1). Few papers were based on human studies (n = 20). Authors reported the results according to biological, metabolic, and histological alterations.
Inflammation
Intentional or accidental peritoneal injury induces a local acute phase reaction, namely, inflammation. Inflammatory response is characterized by cellular infiltration and the release of proinflammatory cytokines such as IL-6, IL-1, and TNF-α. Surgery acts as local trauma and leads to both local and systemic inflammation. Laparoscopy was shown to produce a less severe peritoneal reaction compared to that of an open approach. Therefore, mini-invasive surgery is associated with lower pain and postoperative hospital stay. However, pneumoperitoneum parameters might be modulated during laparoscopy with the aim of reducing early changes related to CO2 insufflation.
In addition to gas type, insufflation pressure was reported to affect peritoneal mucosa integrity and cellular infiltration. In a rat model, low pneumoperitoneum pressure was associated with less intense cellular inflammatory response in terms of eosinophil granulocyte, macrophage, mast-cell, and lymphocyte composition compared to high pneumoperitoneum pressure 24 h after surgery. Therefore, there is a clear relationship between the grade of mucosa inflammation and level of pressure.
Acidosis
Carbon dioxide induces the acidification of fluids, as it has the potential to induce carbonic acid reaction that dissociates in bicarbonate (HCO3-) and hydrogen ions (H+). This phenomenon is well-recognized to occur in blood and cerebrospinal fluid; therefore, several experimental studies investigated the potential ability of CO2 insufflation in inducing peritoneal acidosis. An experimental study in dogs reported that CO2 abdominal insufflation induced acute peritoneal pH reduction after 10–20 minutes. This acidosis was also detectable in the blood, whereas laparotomy was not associated with any pH modification during and after surgery.
Other physical CO2 characteristics were shown to induce pH changes in the abdominal cavity. An experimental swine model compared peritoneal pH in four different groups: standard laparoscopy, laparoscopy with heated and humidified gas, laparoscopy with the administration of heated humidified gas and bicarbonate, and laparotomy. All laparoscopy groups displayed peritoneal acidification, whereas laparotomy did not. The addition of bicarbonate was ineffective in contrasting CO2 acidification. Conversely, the heated and humidified group showed lower pH reduction compared to that of standard laparoscopy. Heating and humidification could enhance local vascularization and subsequently CO2 peritoneal uptake. Nevertheless, the investigation of peritoneal acidosis during laparoscopy is an interesting field of research, as it might be associated with local changes of immune response that could benefit the patient.
Oxidative Stress
During laparoscopy, CO2 insufflation in the abdominal cavity is necessary to visualize the working space; pressure and gas insufflation rate have a pivotal role in this phenomenon. However, increased intra-abdominal pressure might compromise splanchnic perfusion and capillary microcirculation and lead to reduced oxygen content and related ischemia–reperfusion injury with the consequent production of reactive oxygen species (ROS).
ROS release, microcirculation impairment, and tissue hypoxia are the first steps of peritoneal injury, ranging from mesothelial denudation to inflammation and adhesion development. Therefore, efforts in reducing oxidative stress damage might be encouraged to prevent long-term laparoscopy-related comorbidities.
Mesothelial Modifications
The peritoneum is constituted by a single layer of mesothelial cells with an underlying basal membrane and connective tissue containing immune cells, fibroblasts, collagen, and blood vessels. It is involved in the regulation of several biologic processes, such as inflammation, angiogenesis, fibrinolysis, and tissue remodeling. Any kind of mechanical and chemical insult activates inflammation and fibrotic tissue deposition. Therefore, the preservation of the mesothelial lining is crucial for reducing postsurgical adhesions. Animal studies described mesothelium changes during and after a pneumoperitoneum with electron microscopy. This evidence highlights the role of different pneumoperitoneum parameters such as temperature, pressure, flow rate, and humidity in inducing mesothelial damage.
Adhesion Development
Peritoneal adhesions result in the abnormal repair of the peritoneum caused by surgical trauma, inflammation, and infection. Consequently, the peritoneum promotes a healing process covering the damaged tissue with fibrin plug. Normally, the fibrin deposit is entirely removed from the fibrinolytic system, and mesothelial regeneration is achieved in about 8 days. Fibrinolytic activity can be compromised due to ischemic, inflammatory, or thermal insults causing fibroblast proliferation and adherence development.
During laparoscopy, the pneumoperitoneum can cause inflammatory phenomena that result in adhesions in relation to the type, temperature, and pressure of the insufflated gas and the length of pneumoperitoneum.
If considered, the influence of intrabdominal pressure and the perfusion injury, it may be speculated that increased intra-abdominal oxygen tension could be beneficial to the patient.
In the era of minimally invasive surgery, knowledge of the basic physiological mechanisms that are modified by laparoscopy and pneumoperitoneum is essential in making this surgical approach even less invasive. The actor responsible for these changes after laparoscopy is the lining that covers the abdominal cavity, the peritoneum, which maintains an extraordinary balance between all of its components and factors, such as chemokines, cytokines that play an important role in the activation and recruitment of leukocytes to inflammatory sites. Laparoscopy influences both peritoneal integrity and its biology, alters the immune system, and evokes peritoneal acidosis by CO2 insufflation and its influence on microcirculation. It is not clear how all these factors interact with each other and how they can be clinically translated, but knowledge of these mechanisms is the real challenge in truly minimally invasive surgery
Source: Giuseppina Rosaria Umano, Giulia Delehaye, Carmine Noviello, and Alfonso Papparella
Hindawi Minimally Invasive Surgery Volume 2021
https://doi.org/10.1155/2021/5564745
MBBS, MD Obstetrics and Gynecology
Dr Nirali Kapoor has completed her MBBS from GMC Jamnagar and MD Obstetrics and Gynecology from AIIMS Rishikesh. She underwent training in trauma/emergency medicine non academic residency in AIIMS Delhi for an year after her MBBS. Post her MD, she has joined in a Multispeciality hospital in Amritsar. She is actively involved in cases concerning fetal medicine, infertility and minimal invasive procedures as well as research activities involved around the fields of interest.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


