- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Subconjunctival dexamethasone implant lowers risk of graft rejection after penetrating keratoplasty
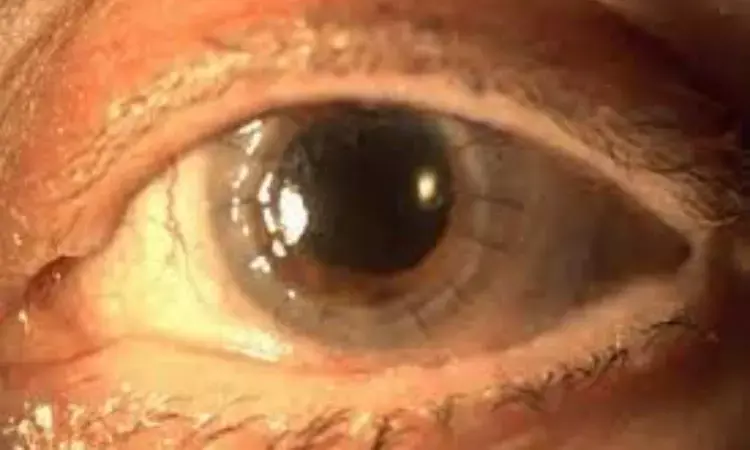
Subconjunctival dexamethasone implant may be a safe and well-tolerated immunosuppressive therapy for patients after low-rejection risk penetrating keratoplasty suggests a new study published in the British Journal of Ophthalmology
Rejection is the main cause of graft failure after penetrating keratoplasty (PK). Its prevention by repeated instillation of steroid eye-drops has not evolved in decades. Poor adherence and discontinuous nature of eye-drop treatment may explain some PK failures. In a rabbit model, we previously demonstrated that a subconjunctival dexamethasone implant was well tolerated and prevented rejection efficiently in the first 5-6 weeks. This clinical trial investigates its tolerance and safety after PK.
Single-centre, phase II non-randomised tolerance and safety pilot study (NCT02834260). Designed to analyse the risk of elevated intraocular pressure (IOP), discomfort and resorption time. Fourteen patients with a low rejection risk indication of PK were enrolled between January 2017 and August 2018. The implant was injected in the 12 o'clock position, 5 mm from the limbus, at the end of PK. A steroid eye-drop treatment was planned when implant resorption was complete. Patients were monitored regularly for 12 months: IOP (main outcome measure at 1 month), discomfort and redness scores, implant status, rejection episode and central corneal thickness by optical coherence tomography. An independent data safety monitoring committee verified safety aspects.
Results:
No increase in IOP or other adverse event related to the implant was observed. Average resorption time was 6 weeks. The switch to steroid eye-drops was uneventful. One patient, included despite preoperative corneal neovascularisation (unintended protocol deviation) experienced a rejection.
This is the first proof of concept that dropless immunosuppression is possible after low rejection risk PK.
Reference:
Trone MC, Poinard S, Crouzet E, Garcin T, Mentek M, Forest F, Matray M, Thuret G, Gain P. Dropless penetrating keratoplasty using a subconjunctival dexamethasone implant: safety pilot study. Br J Ophthalmol. 2023 Feb;107(2):181-186. doi: 10.1136/bjophthalmol-2021-319376. Epub 2021 Aug 23. PMID: 34426402.
Keywords:
British Journal of Ophthalmology, Subconjunctival, dexamethasone, implant, safe, well-tolerated immunosuppressive, therapy, patients, low-rejection, risk, PK, Trone MC, Poinard S, Crouzet E, Garcin T, Mentek M, Forest F, Matray M, Thuret G, Gain P.
Dr. Shravani Dali has completed her BDS from Pravara institute of medical sciences, loni. Following which she extensively worked in the healthcare sector for 2+ years. She has been actively involved in writing blogs in field of health and wellness. Currently she is pursuing her Masters of public health-health administration from Tata institute of social sciences. She can be contacted at editorial@medicaldialogues.in.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


