- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Use of Povidone Iodine for Endophthalmitis Prophylaxis linked to Iodine Allergy
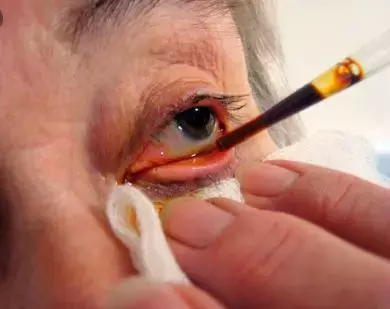
Iodine is a necessary element for life, thus an allergy to elemental iodine is not possible. Nevertheless, iodine containing chemicals can provoke 3 types of reactions on human contact: an irritant contact dermatitis, an allergic contact dermatitis, and a generalized anaphylaxis.
First, an irritant reaction to iodine is an effect of the body's innate immune system and, as such, a skin response can be provoked immediately without prior exposure. Compared with tinctures of pure iodine, these types of reactions are less frequent with the lower levels of free iodine encountered with iodophores, but they are still common and depend on the exposure time and concentration. As opposed to an irritant reaction, an allergic reaction to the iodophore is mediated by T lymphocytes.
In a review published in The Journal of Vitreoretinal Diseases ,authors have discussed the role of povidone-iodine (PI) for endophthalmitis prophylaxis in ophthalmic procedures, stating that it is an essential component, that true PI allergy is rare, and that reported allergy to iodine, shellfish, or contrast media is not a contraindication.
It is now well documented that the antiseptic povidone iodine (PI) is a key component of endophthalmitis prophylaxis. Due to wide evidence of data showing that PI effectively reduces bacterial counts in 30 seconds and that the fastest in vitro killing time is achieved with 0.1% PI rather than more-concentrated formulations, The American Academy of Ophthalmology recommends using 5% PI,25 and recent in vivo studies have shown that 5% PI is as effective as 10% in the setting of IVIs.
"Despite its availability, 34 years passed before the significant utility of PI was demonstrated in a clinical trial of cataract surgery. it is now standard practice not only in the operating room but also in intravitreal injection (IVI) protocols.6-8 Key aspects of PI include short application time for effectiveness,9 efficacy of dilute concentrations,10 lack of microbial resistance,11,12 ability to penetrate biofilm,13 and safety to the retina." wrote the reviewers.
According to the team, a recent retrospective review emphasized the increased rate of endophthalmitis in IVIs without PI and the much lower risk when it is used. This study reported that 11.9% of post-IV endophthalmitis cases occurred when PI was omitted from the preparation. Furthermore, injections lacking PI antisepsis had an estimated infection rate of 9.4%, whereas the study's overall incidence of endophthalmitis after injection was 0.019%. These findings emphasize that, despite its critical, widely accepted role in endophthalmitis prevention, a clinical dilemma arises when patients endorse an "allergy" that may contraindicate PI application.
The reviewers further stated that though present guidelines note that true allergy to PI is rare and anaphylaxis after ophthalmic use has never been described, physicians still hesitate to use the antiseptic in these situations. Although patients may have true allergies to these other substances, no data suggest that using topical PI for endophthalmitis prophylaxis in these patients is contraindicated.
"PI is an essential component of reducing the risk of endophthalmitis after ophthalmic procedures. As such, clinical scenarios that justify withholding PI are rare. Given the infrequency of a true allergy to PI, physicians should critically consider any reported reactions to the antiseptic itself."concluded the reviewers.
Primary source: Journal of Vitreoretinal Diseases
For full article click on the link: "Iodine Allergy" and the Use of Povidone Iodine for Endophthalmitis Prophylaxis
J Vitreoretin Dis 2019 Oct 14;4(1)65, JW Hinkle, CC Wykoff, JI Lim, P Hahn, SJ Kim, H Tabandeh, HW Flynn Jr
Dr Satabdi Saha (BDS, MDS) is a practicing pediatric dentist with a keen interest in new medical researches and updates. She has completed her BDS from North Bengal Dental College ,Darjeeling. Then she went on to secure an ALL INDIA NEET PG rank and completed her MDS from the first dental college in the country – Dr R. Ahmed Dental College and Hospital. She is currently attached to The Marwari Relief Society Hospital as a consultant along with private practice of 2 years. She has published scientific papers in national and international journals. Her strong passion of sharing knowledge with the medical fraternity has motivated her to be a part of Medical Dialogues.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


