- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
FDA approves RemeOs trauma screw as first bioresorbable metal implant for marketing
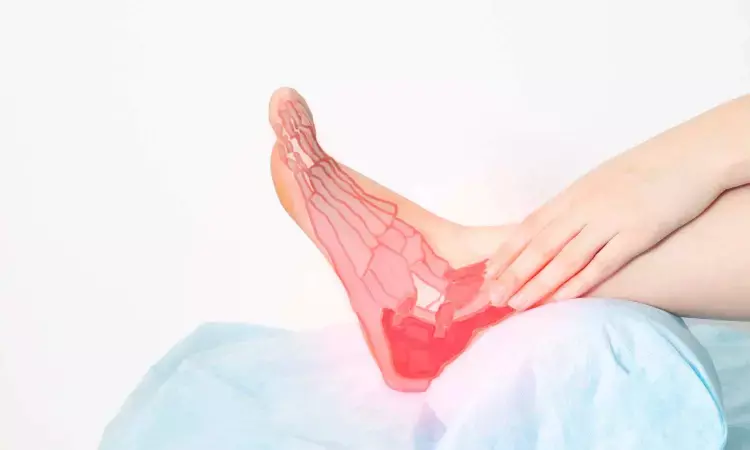
The Food and Drug Administration has granted market authorization to Bioretec for its RemeOs trauma screw which is designed to aid fixation for patients with ankle fractures.
The benefits of the RemeOs trauma screw have been validated in clinical trials and have already previously been recognized by the FDA granting the Breakthrough Device Designation. Bioresorbable metals combine the surgical techniques of traditional metal implants and the patient-friendly care and benefits of last-generation bioresorbable polymer implants making implant removal operations redundant.
"This FDA market approval is the most important milestone in our company's history to date,” comments Timo Lehtonen, CEO of Bioretec.“ The U.S. has the world's largest market for orthopedic implants. For the first time, that market is now offered a bioresorbable alternative to be used instead of titanium and steel implants. We are confident that RemeOs trauma screw is the best option for a large share of patients considering the care and clinical outcome and from a health economics viewpoint thanks to making an additional removal operation unnecessary. We are also excited about this approval opening a less burdensome regulatory pathway for future RemeOs product lines covering a wide array of indications."
Ankle fractures are one of the most frequently occurring fracture types among the adult patient population. There are 3.4 million patients treated each year in the U.S. for ankle fractures. Single-isolated ankle (malleolar) fractures are the most common type, accounting for 70% of the yearly incidence of all ankle fractures. The U.S. ankle and foot market is estimated to grow (CAGR) by 7.3% per year. Bioretec will launch the RemeOs trauma screws in the U.S. in collaboration with several hospitals and clinical professionals specialized in ankle fractures. The company expects that RemeOs sales will grow gradually as the clinics gain confidence in the innovative bioresorbable metal implant, as is customary for all new orthopedic products.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


