- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Azithromycin has no effect on growth and development of children: JAMA
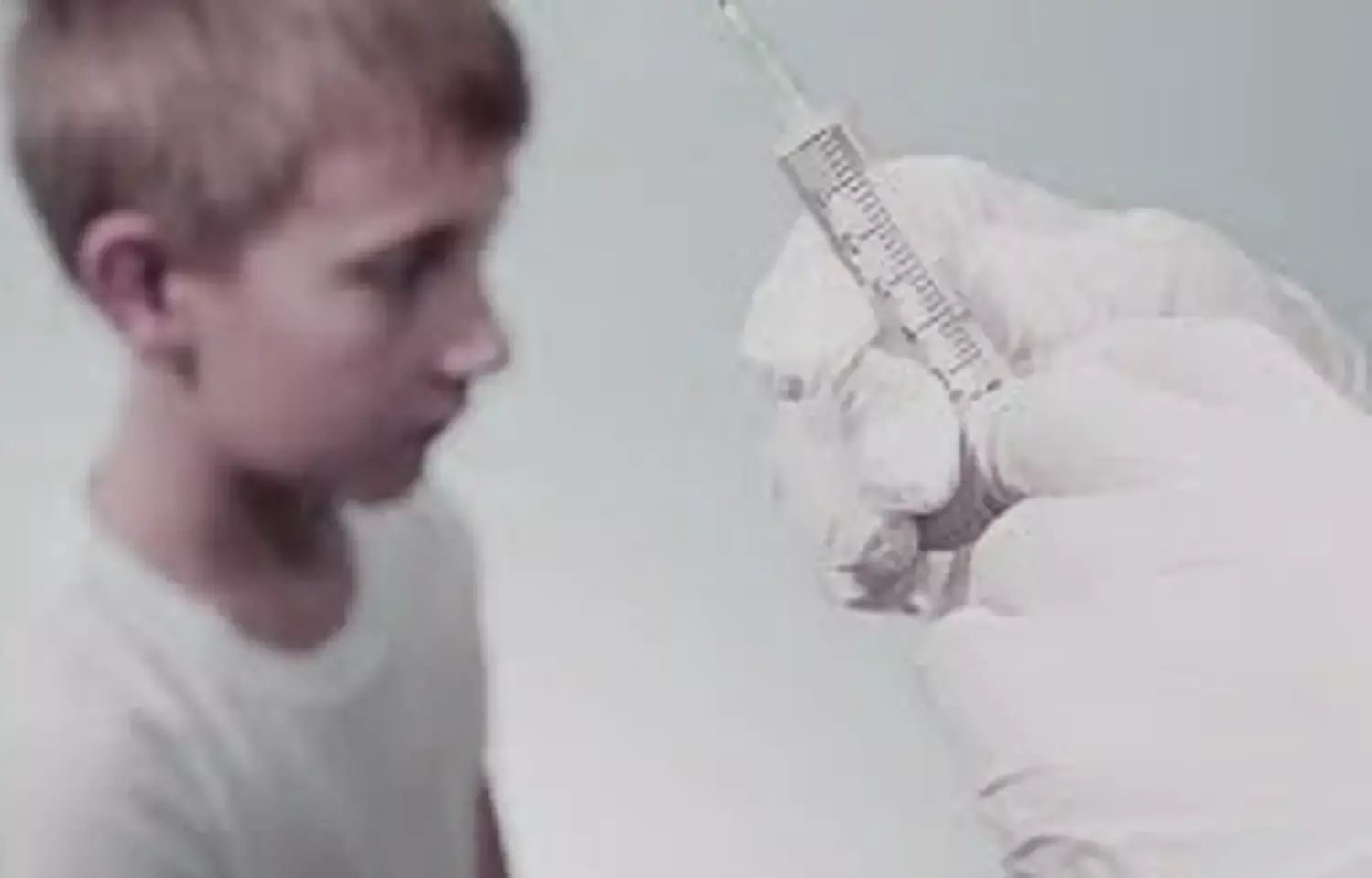
Although the exact mechanism is unknown, mass azithromycin deployments may reduce childhood mortality. Some of the mortality benefits may be explained by antibiotics' ability to act as growth promoters.
A recent research revealed no indication of a link between mass azithromycin distributions and kid development, however subgroup analyses indicated some advantage for the shortest children.
This study was conducted by Ahmed M. Arzika and team with the objective To see if biennial bulk azithromycin distributions are linked to improved childhood growth. The findings of this study were published in the Journal of American Medical Association on 30th December, 2021.
From December 2014 to March 2020, this cluster-randomized experiment was conducted in 30 rural towns in Niger's Boboye and Loga departments, with populations ranging from 200 to 2000 people. For children aged 1 to 59 months, communities were randomized in a 1:1 ratio to biennial mass distributions of azithromycin or placebo. Treatment allocation was concealed from participants, field workers, and research personnel. The changes in height and weight from baseline to the 4-year follow-up were compared across groups.
The data was evaluated from June to November 2021. Every 6 months, participants' homes got azithromycin at a dose of 20 mg/kg depending on height or weight for youngsters unable to stand. The vehicle for the azithromycin suspension was contained in the placebo. Longitudinal anthropometric assessments were performed on a random sample of children before the first treatment and then annually for 5 years. Height and weight were the prespecified primary outcomes.
The key findings of this study were:
1. The baseline characteristics of 1299 children in the azithromycin group and 2637 children in the placebo group were similar among 3936 children enrolled from 30 areas.
2. Baseline anthropometric evaluations were done on 2230 children, 985 in the azithromycin group and 1245 in the placebo group, with follow-up measures available for 789 (80.1%) and 1063 (85.4%), respectively.
3. Children in the azithromycin group grew a mean of 6.7 cm in height and 1.7 kg in weight each year at the 4-year follow-up visit, whereas children in the placebo group gained a mean of 6.6 cm in height and 1.7 kg in weight per year.
4. Height at 4 years was not statistically different across groups when corrected for baseline height, and weight was not statistically different when adjusted for height and baseline weight.
5. However, as compared to placebo, azithromycin was related with a 0.4 cm gain in height among children in the smallest percentile of baseline height.
In conclusion, This study discovered that biennial mass azithromycin distributions had no significant effect on growth rates in a placebo-controlled cluster-randomized trial in Niger, however subgroup analysis revealed a possible advantage among children in the shortest quartile. These data imply that the decrease in mortality transmitted by bulk azithromycin distributions is most likely not due to growth promotion alone.
Reference:
Arzika AM, Maliki R, Ali MM, et al. Effect of Mass Azithromycin Distributions on Childhood Growth in Niger: A Cluster-Randomized Trial. JAMA Netw Open. 2021;4(12):e2139351. doi:10.1001/jamanetworkopen.2021.39351
Medical Dialogues consists of a team of passionate medical/scientific writers, led by doctors and healthcare researchers. Our team efforts to bring you updated and timely news about the important happenings of the medical and healthcare sector. Our editorial team can be reached at editorial@medicaldialogues.in.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


