- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Tofacitinib may induce hair regrowth in over 80 percent children with alopecia: Study
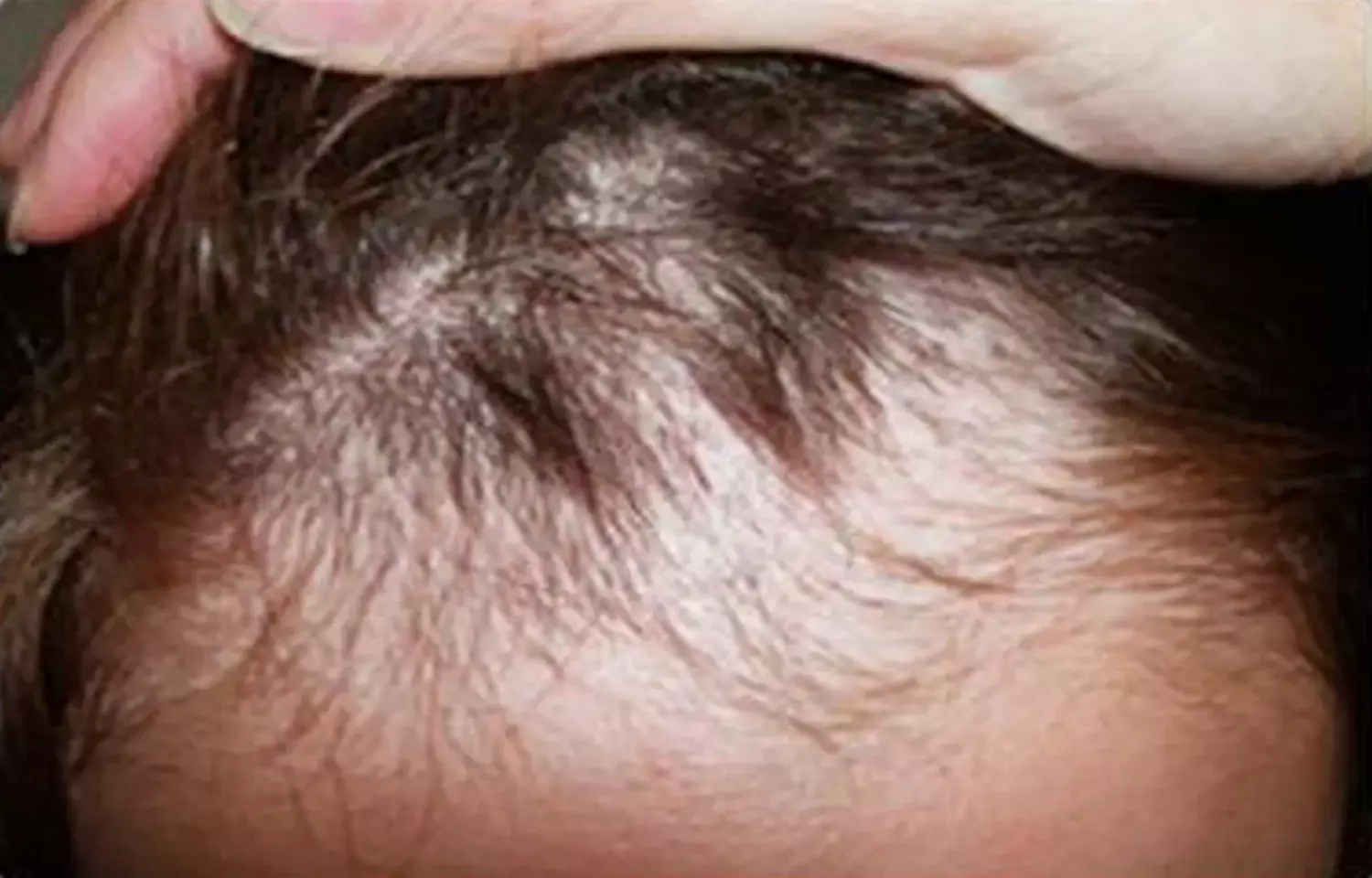
Australia: Oral tofacitinib may be an effective and well-tolerated treatment strategy for in some children with alopecia areata (AA), suggests a recent study in the journal Pediatric Dermatology.
Tofacitinib, a selective 1/3 janus kinase inhibitor (JAKi), is used for the treatment of inflammatory diseases including alopecia areata, vitiligo, sarcoidosis, systemic lupus, atopic dermatitis, psoriasis, rheumatoid arthritis, and psoriatic arthritis. Alopecia areata is an autoimmune hair loss condition that affects people across all age groups. Prolonged disease duration and early age of onset is linked to poor prognosis. Phase 3 trials are investigating the role of janus kinase inhibitors in adults and adolescents with AA.
Rebekka Jerjen, Sinclair Dermatology, Melbourne, VIC, Australia, and colleagues evaluated the use of oral tofacitinib in pre‐adolescent patients with AA.
For the purpose, they performed a retrospective review of case records of all pre‐adolescent patients having AA and treated with oral tofacitinib in a single center between 2018 and 2019. 14 patients aged 7 to 11 years were identified.
Key findings of the study include:
- Nine patients experienced clinically significant improvement in their SALT (Severity of Alopecia Tool) score.
- Three patients achieved complete remission (SALT score of 0), seven (63.6%) achieved over 50% improvement in SALT score from baseline.
- One patient had no change from baseline, another experienced additional hair loss.
- After an average of 9 months of treatment, the median SALT score improvement was 67.7%.
- The improvement was similar in patients with baseline SALT scores greater than 50 and those with baseline SALT scores below 10.
- Adverse events were mild.
To sum up, in this study tofacitinib was well tolerated, 63.6% of patients experienced over 50% improvement in their SALT scores and 82% of patients experienced some hair regrowth.
"The results indicate a role for tofacitinib as a systemic therapy in AA in the pre-adolescent population and corroborate findings from prior larger studies in adolescents and adults. Larger prospective studies to ensure long-term safety with use of JAKi in children, as well as patient-reported qual-ity of life assessments, are still required," concluded the authors.
"Treatment of alopecia areata in pre‐adolescent children with oral tofacitinib: A retrospective study," is published in the journal Pediatric Dermatology.
DOI: https://onlinelibrary.wiley.com/doi/abs/10.1111/pde.14422
MSc. Biotechnology
Medha Baranwal holds a Bachelor’s degree in Biomedical Sciences from the University of Delhi and a Master’s degree in Biotechnology from Amity University. Since May 2018, she has been contributing to Medical Dialogues, writing and editing medical news articles that translate complex research into clear, accessible information for healthcare professionals.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


