- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
FDA clears microwave ablation system for removing soft tissues like thyroid nodules
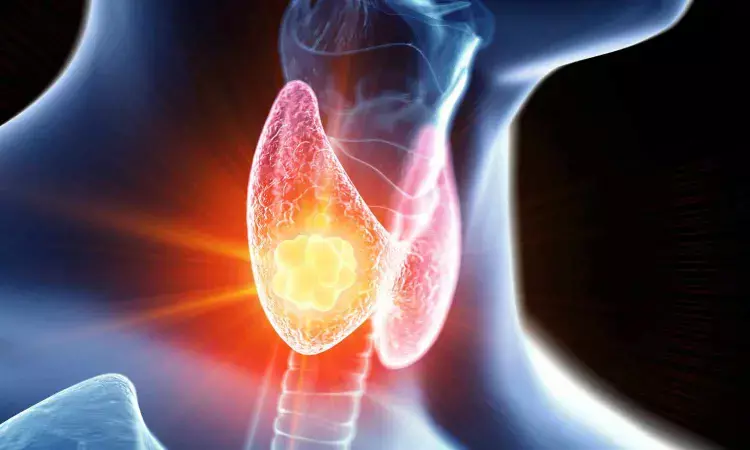
The US Food and Drug Administration has cleared a microwave ablation system for removing soft tissues like thyroid nodules.
Thyroid nodules occur among 30% to 50% of the United States population. Like most thyroid conditions, nodules are more common among women than men, with an increased incidence observed with advancing age. By the age of 60, more than half of women will have developed a thyroid nodule. Historically, treatment involving excision or therapy resulted in discomfort and inconvenience for patients. In contrast to these traditional approaches, Baird Medical has leveraged significant recent technological advances to develop a minimally invasive, thermal energy-based procedure (the "Procedure") to treat thyroid nodules, breast tumors, and other diseases for which the coagulation (ablation) of soft tissues is indicated. The Company's approach eliminates the need for surgery, thereby mitigating associated complications, risks, and scarring for the majority of patients.
During the Procedure, therapeutic microwave energy is safely administered via a device approximately the diameter of a pen. This minimally invasive treatment precisely targets the nodule or tumor being treated, while preserving the surrounding healthy tissue. This precision is crucial, as it safeguards the function of the thyroid and reduces the likelihood that a patient will require expensive, lifelong thyroid hormone replacement therapy.
In light of the increasing scientific evidence demonstrating positive outcomes associated with the expanded availability of the Procedure, both the American Thyroid Association and the European Thyroid Association have published recommended guidelines for the implementation and adoption of the Procedure. Physician associations, such as The North America Society for Interventional Thyroidology, have been established to raise awareness among physicians and patients that a minimally invasive solution exists for treating thyroid disease.
Ms. Haimei Wu, Founder and CEO of Baird Medical, commented, "Attaining FDA clearance represents a significant milestone in our geographical expansion. This clearance grants us the ability to market our portfolio of MWA systems and disposable needles in the United States. Based on our market research, we estimate that the U.S. presents a market opportunity of $1.6 billion for MWA systems alone. Because our follow-on sales of disposable MWA needles typically generate 36 times the revenue of our initial MWA system sales, we anticipate a total addressable market size of $1.6 billion in the U.S. alone. We are excited about our potential to capitalize on such a vast market opportunity. Our team of skilled professionals is ready to expand our market reach and deliver the much-needed benefits of our minimally invasive MWA procedure to patients across America."
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


