- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Pulmonary artery denervation shows promise for group I PAH patients, JACC study.
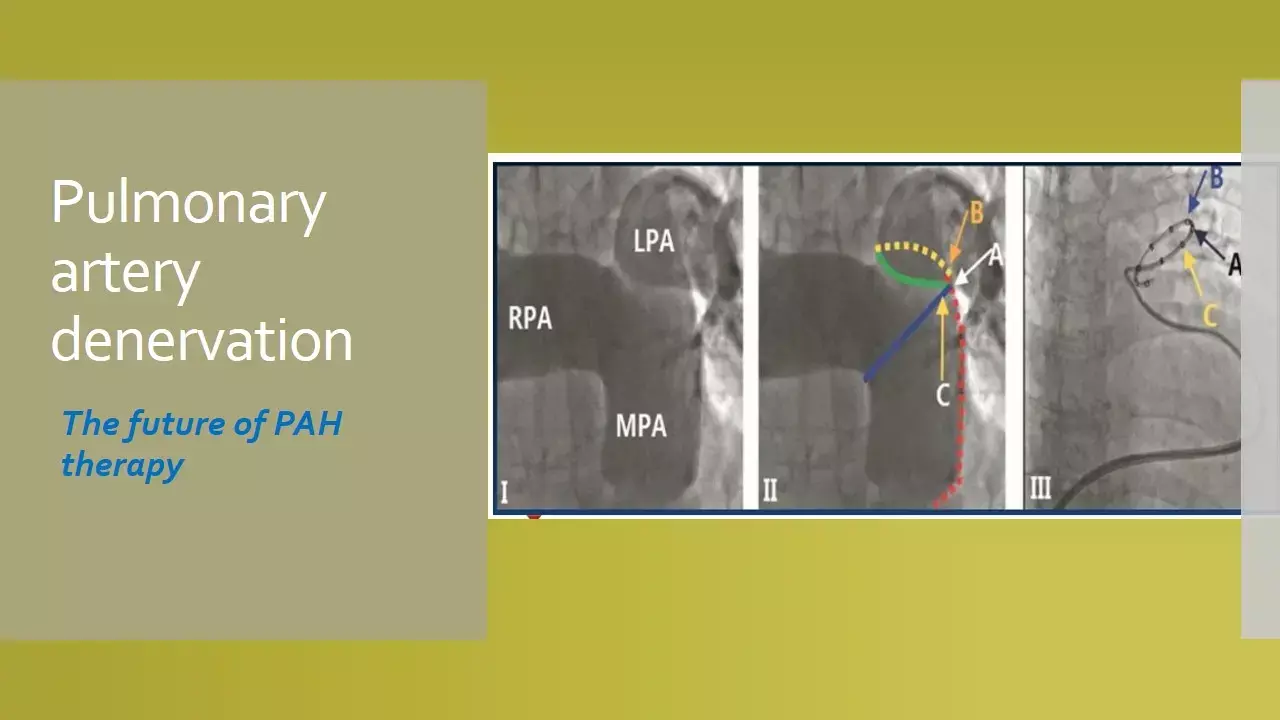
WHO Group I pulmonary arterial hypertension (PAH) is a progressive, debilitating disease. Previous observational studies have demonstrated that pulmonary artery denervation (PADN) reduces pulmonary arterial pressures in PAH. However, the safety and effectiveness of PADN have not been established in a randomized trial.
In this regard, the results of a randomised sham-controlled published recently in JACC CI, have shown that in patients with WHO Group I PAH, PADN improved exercise capacity, hemodynamics and clinical outcomes during 6-month follow-up.
While advances in medical therapies have improved symptom control and clinical outcomes, patients with PAH have a poor long-term prognosis. A hyperactive sympathetic nervous system has been implicated in PAH.
In an early human study PADN reduced pulmonary arterial pressures and improved exercise capacity during 3-month follow-up. A subsequent study demonstrated improved hemodynamics, exercise performance, and cardiac function 1-year after PADN. Authors Zhang et al therefore performed a multicenter, randomized, single-blind trial to evaluate the safety and effectiveness of PADN in PAH.
WHO Group I PAH patients not taking PAH-specific drugs for at least 30 days were enrolled in a multicenter, sham-controlled single-blind, randomized trial. Patients were assigned to receive PADN plus a phosphodiesterase-5 inhibitor (PDE-5i) or a sham procedure plus a PDE-5i. The primary endpoint was the between-group difference in the change in 6-minute walk distance (6MWD) from baseline to 6 months.
1. Among 128 randomized patients, those treated with PADN compared with sham had a greater improvement in 6MWD from baseline to 6.
2. From baseline to 6 months PVR was reduced by -3.0 ± 0.3 Wood units after PADN and -1.9 ± 0.3 Wood units after sham. PADN also improved right ventricular function, reduced tricuspid regurgitation and decreased NT-proBNP.
3. Clinical worsening was less and a satisfactory clinical response was with PADN treatment during 6-month follow-up.
No major complications occurred from the PADN procedure, similar to previous studies. The incremental improvement in 6MWD with PADN (median 33.8 m) in patients on the background PDE-5i treatment was at least as good as in prior trials in which a second PAH-specific drug was added to monotherapy, or a third drug was added to combination therapy.
Since follow-up for only 6 months is available at present, hence longer-term assessments (planned for 3 years) are essential to examine the durability of the present outcomes.
To summarize, in the present sham-controlled, single-blind randomized trial of stable patients with PAH, treatment with PADN plus a PDE-5i was safe and resulted in improved exercise capacity at 6 months compared with background therapy of PDE-5itreatment alone.
Source: JACC CI: DOI: 10.1016/j.jcin.2022.09.013
MBBS, MD , DM Cardiology
Dr Abhimanyu Uppal completed his M. B. B. S and M. D. in internal medicine from the SMS Medical College in Jaipur. He got selected for D. M. Cardiology course in the prestigious G. B. Pant Institute, New Delhi in 2017. After completing his D. M. Degree he continues to work as Post DM senior resident in G. B. pant hospital. He is actively involved in various research activities of the department and has assisted and performed a multitude of cardiac procedures under the guidance of esteemed faculty of this Institute. He can be contacted at editorial@medicaldialogues.in.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


