- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
TTVR Tied to Safe and Effective Real-World Outcomes in Severe TR, Finds Study
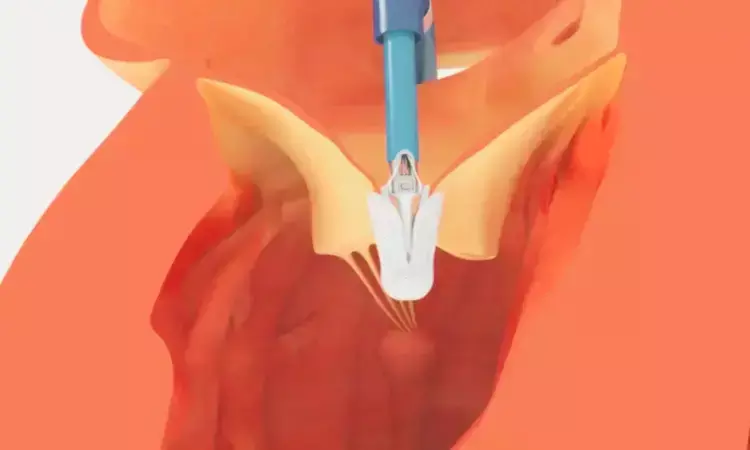
USA: Researchers have discovered in a new research that transcatheter tricuspid valve replacement (TTVR) is safe and effective for patients with severe tricuspid regurgitation (TR). Thirty-day outcomes align with the TRISCEND II trial, showing near-complete TR reduction and improved health status, with lower rates of device implantation and bleeding compared to clinical trials.
- Successful valve implantation was achieved in 98.4% of patients, indicating high procedural success.
- Mild or less tricuspid regurgitation was observed in nearly all patients post-procedure and in 97.7% at 30 days.
- Findings demonstrate near-complete elimination of regurgitation in routine clinical practice.
- At 30 days, all-cause mortality was 3.1% and stroke incidence was 0.2%.
- Bleeding events occurred in 7.9% of patients.
- Heart failure hospitalizations were reported in 3.1% of patients.
- Among patients without prior devices, 15.9% required new cardiac implantable electronic device placement.
- Overall safety outcomes were acceptable and in some cases lower than those seen in clinical trials.
- Significant improvement in functional status was observed, with over 80% of patients in NYHA class I or II at 30 days.
- Quality of life improved markedly, with an increase of more than 22 points in KCCQ score.
- Results indicate substantial symptom relief and better daily functioning.
- Baseline presence or absence of cardiac implantable electronic devices did not affect short-term mortality.
- No significant differences were observed in heart failure hospitalization or functional outcomes across subgroups.
- The benefits of TTVR were consistent across different patient groups.
MSc. Biotechnology
Medha Baranwal holds a Bachelor’s degree in Biomedical Sciences from the University of Delhi and a Master’s degree in Biotechnology from Amity University. Since May 2018, she has been contributing to Medical Dialogues, writing and editing medical news articles that translate complex research into clear, accessible information for healthcare professionals.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


