- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Dupilumab Improves Itch and Disease Activity in Chronic Spontaneous Urticaria Regardless of BMI, Gender: Study
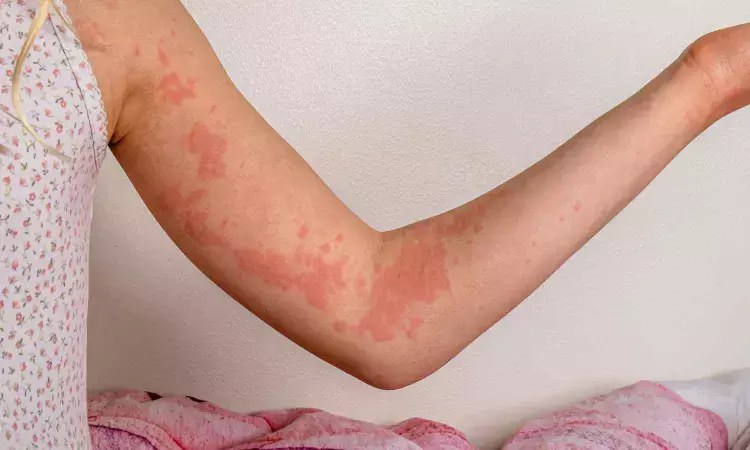
USA: A study published in The Journal of Allergy and Clinical Immunology reports that Dupilumab significantly reduced itch severity and disease activity in patients with Chronic Spontaneous Urticaria (CSU), with benefits observed consistently across different patient subgroups. The analysis indicates that factors such as body mass index (BMI), gender, and the presence of angioedema did not substantially alter the treatment response.
- After 24 weeks of treatment, patients receiving Dupilumab showed greater improvement in itch severity compared with the placebo group.
- Reductions in the 7-day Itch Severity Score (ISS7) were observed across all body mass index (BMI) categories, including patients with normal weight, overweight, and obesity.
- Improvements in itch severity were consistent regardless of the presence or absence of angioedema at baseline.
- Both male and female patients experienced similar improvements in itch severity with dupilumab therapy.
- Greater reductions in the 7-day Urticaria Activity Score (UAS7) were also observed in patients treated with dupilumab compared with placebo.
- Improvements in urticaria activity were consistent across different BMI groups, angioedema status, and gender.
- The safety profile of dupilumab was consistent with previously reported data.
- No new or unexpected safety concerns were identified during the study period.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


