- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
FDA extends approval of Dupilumab for Young Children with Chronic Spontaneous Urticaria
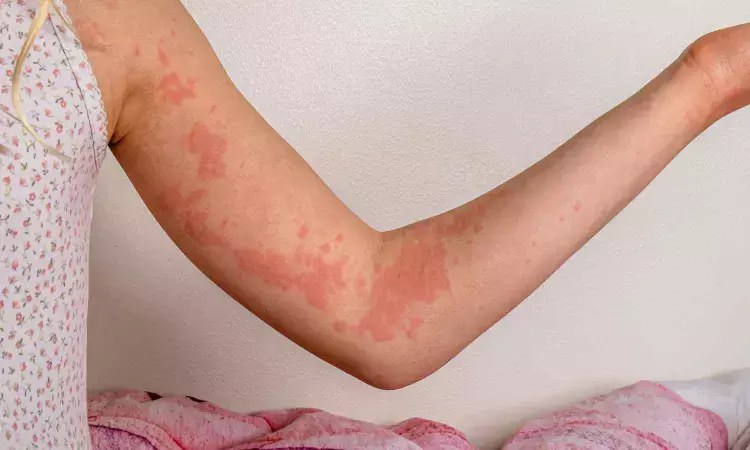
The FDA has expanded approval of dupilumab to include children aged 2 to 11 years with chronic spontaneous urticaria who continue to have symptoms despite H1 antihistamine therapy. This decision is supported by evidence from the LIBERTY-CUPID clinical trial program, demonstrating its efficacy in this pediatric population.
This expands the previous approval for Dupixent in adults and adolescents aged 12 years and older with CSU.
“Children with uncontrolled chronic spontaneous urticaria continue to experience the unpredictable appearance of debilitating itch and hives,” said Alyssa Johnsen, MD, PhD, Global Therapeutic Area Head, Immunology Development at Sanofi. “Until now, these patients had to rely on limited treatment options that didn’t address potential critical mediators of chronic spontaneous urticaria. Dupixent is the first biologic approved for patients as young as two years of age, offering a targeted approach that inhibits IL4 and IL13 signaling, two key and central drivers of the type 2 inflammation that contributes to this disease. Today’s approval underscores our ongoing commitment to advancing therapies for young patients with significant unmet needs.”
The approval is based primarily on data from the LIBERTY-CUPID clinical study program. This includes extrapolation of efficacy and safety data from two phase 3 studies (Study A and Study C; clinical study identifier: NCT04180488) in certain adults and adolescents aged 12 years and older with CSU complemented with pharmacokinetics data from the single-arm, CUPIDKids (clinical study identifier: NCT05526521) phase 3 study in children aged 2 to 11 years with CSU. In Study A and Study C, Dupixent significantly reduced itch severity and urticaria activity (a composite of itch and hives) compared to placebo at Week 24. In adults and adolescents, Dupixent also increased the likelihood of well-controlled disease or complete response compared to placebo at Week 24. Study B (clinical study identifier: NCT04180488) provided additional safety data and evaluated Dupixent in patients aged 12 years and older who were inadequate responders or intolerant to anti-IgE therapy and symptomatic despite antihistamine use. Safety in children aged 2 to 11 years with CSU was supported by data from pediatric patients in other indications.
The safety results from all four CSU studies were generally consistent with the known safety profile of Dupixent in its approved dermatological indications. In Study A, Study B, and Study C, the most common adverse reaction (≥2%) in the US Prescribing Information more frequently observed in patients on Dupixent compared to placebo was injection site reactions. No new adverse reactions were identified in children aged 2 to 11 years with CSU treated with Dupixent.
“With this approval, Dupixent has become the first biologic medicine in the US for young children suffering from uncontrolled chronic spontaneous urticaria, an unpredictable skin disease that impacts quality of life during these children’s most formative years,” said George D. Yancopoulos, MD, PhD, Board co-Chair, President and Chief Scientific Officer at Regeneron. “Dupixent is now approved for nine different allergy-related conditions, from asthma to atopic dermatitis, and this is the fifth of these indications now extended to young children. The FDA’s authorization reinforces our medicine’s well-established safety profile and potential to transform outcomes for chronic diseases driven in part by type 2 inflammation impacting some of the most vulnerable populations. As the most widely used innovative branded antibody medicine, Dupixent, has the potential to change yet another treatment paradigm.”
In addition to the US, Dupixent is approved for CSU in certain children aged two to 11 years in the EU and other countries around the world.
About CSU
CSU is a chronic inflammatory skin disease driven in part by type 2 inflammation, which causes sudden and debilitating hives and recurring itch. CSU is typically treated with H1AH, medicines that target H1 receptors on cells to control symptoms of itch and urticaria. However, the disease remains uncontrolled despite H1AH treatment in more than 14,000 children in the US aged two to 11 years living with CSU, some of whom are left with limited alternative treatment options. These individuals continue to experience symptoms that can be debilitating and significantly impact their quality of life.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


