- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Female Androgenetic Alopecia effectively treated by PRP, finds study
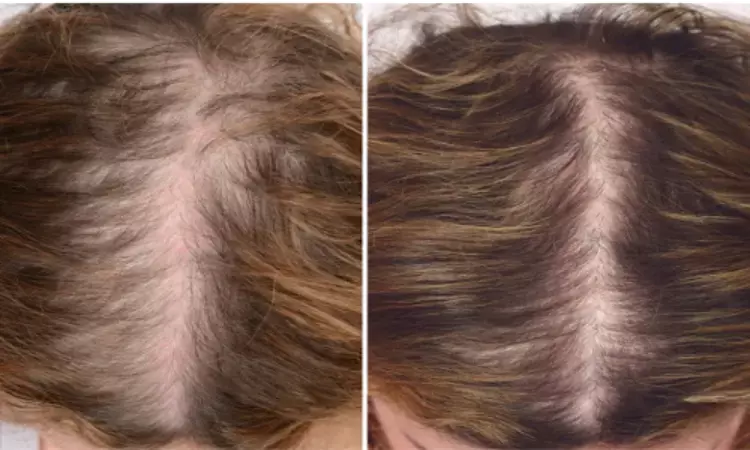
The eclipse system Platelet rich plasma (PRP) is found to be a safe and effective intervention in the treatment of Androgenetic Alopaecia, finds a recent trial done in the New York. The study was published in the Journal of the American Academy of Dermatology, 2020.
Androgenetic Alopecia (AGA) which is a female pattern of hair loss can cause considerable distress both emotionally and psychologically and can even decrease the quality of life. It is a non scarring type of diffuse alopecia which occurs due to genetic and hormonal factors. Apart from the regular standard treatment protocols, PRP has gained much popularity in the recent times due to its less side effects and cost affordability.
Also Read: Low dose Oral Minoxidil Safe for Non-scarring Alopecia, finds study
The researchers from the School of Medicine, New York conducted a prospective randomized controlled trial to determine whether PRP injections improve female AGA. 30 females diagnosed with AGA were included in the trial but 2 patients were lost to follow up. Patients received sub-dermal scalp injections of Eclipse system PRP or placebo saline at weeks 0, 4 and 8. Outcome measures were changes in hair density (hair/cm2), hair caliber (mm), and blinded global photographic assessment (improved or not improved) at week 24.
The fey findings of the study were:
• Blinded global photographic assessment indicated that 57% of PRP patients vs. 7% of saline patients improved at week 24 from baseline (p<0.01).
• Compared to baseline, there was improvement in mean density in the PRP group vs. the placebo group at week 8 (+71.1 vs. -26.7 hairs/cm2, p<0.01) and week 24 (+105.9 vs. -52.4 hairs/cm2, p<0.01).
• Compared to baseline, there was improvement in mean caliber in the PRP group vs. the placebo group at week 8 (+0.0043 vs. -0.0034 mm, p<0.01) and week 24 (+0.0053 vs. -0.0060 mm, p<0.01).
• Adverse effects included headache, scalp tightness, swelling, redness, and post-injection bleeding.
Thus, the researchers concluded that PRP with the Eclipse system is a safe and effective intervention for female AGA.
To read more about the study, click the following link: https://doi.org/10.1016/j.jaad.2020.06.1021
BDS, MDS
Dr.Niharika Harsha B (BDS,MDS) completed her BDS from Govt Dental College, Hyderabad and MDS from Dr.NTR University of health sciences(Now Kaloji Rao University). She has 4 years of private dental practice and worked for 2 years as Consultant Oral Radiologist at a Dental Imaging Centre in Hyderabad. She worked as Research Assistant and scientific writer in the development of Oral Anti cancer screening device with her seniors. She has a deep intriguing wish in writing highly engaging, captivating and informative medical content for a wider audience. She can be contacted at editorial@medicaldialogues.in.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


