- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Study Highlights Long-Term Efficacy of Ritlecitinib in Alopecia Areata
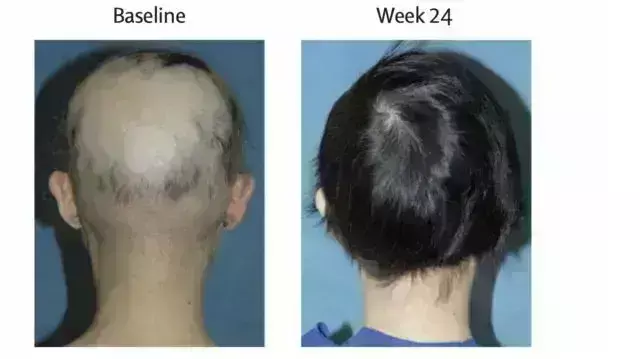
Research suggests that ritlecitinib is effective for long-term treatment in patients with alopecia areata who have greater than or equal to 25% scalp hair loss. The study found that patients achieved significant hair regrowth and reported notable improvements in their condition. These findings emphasize the effectiveness of prolonged ritlecitinib treatment for individuals with significant scalp hair loss due to alopecia areata.
ALLEGRO-LT is an ongoing, long-term, open-label, multicentre, phase 3 study of ritlecitinib in adults and adolescents with alopecia areata (AA). A study was done to evaluate ritlecitinib safety and efficacy through Month 24 in patients with AA and ≥25% scalp hair loss. ALLEGRO-LT enrolled rollover patients who previously received study intervention in either ALLEGRO phase 2a or 2b/3 studies and de novo patients who had not received treatment in either study. The de novo cohort results are reported here. Patients aged ≥12 years with AA and ≥25% scalp hair loss received a daily, 4-week 200-mg ritlecitinib loading dose, followed by daily 50-mg ritlecitinib. Analyses are based on data up to the cut-off (December 2022). Efficacy outcomes included proportions of patients achieving Severity of Alopecia Tool (SALT) scores ≤20 and ≤10, Patient Global Impression of Change (PGI-C) score of 'moderately improved' or 'greatly improved' and eyebrow assessment (EBA) and eyelash assessment (ELA) response (≥2-grade improvement from baseline or normal score in patients with abnormal baseline EBA/ELA). Results: Mean (SD) ritlecitinib exposure among the 449 de novo patients enrolled was 728.7 (273.81) days. At Month 24 (as observed), 73.5% and 66.4% of patients achieved SALT score ≤20 and ≤10; 82.4% had PGI-C response; 60.8% and 65.7% had EBA and ELA response. 86.1% of patients reported treatment-emergent adverse events (AEs); most were mild or moderate in severity, with the most frequent being positive SARS-CoV-2 test (24.2%), headache (20.8%) and pyrexia (13.0%). Rates of serious AEs, severe AEs and treatment discontinuations were 4.9%, 6.0% and 6.5%, respectively. Herpes zoster infection occurred in six patients, serious infections in four, malignancies (excluding nonmelanoma skin cancer) in three and major adverse cardiovascular events in three. In patients with AA and ≥25% scalp hair loss, ritlecitinib demonstrated clinical efficacy and had an acceptable safety profile with long-term treatment.
Reference:
Tziotzios C, Sinclair R, Lesiak A, Mehlis S, Kinoshita-Ise M, Tsianakas A, Luo X, Law EH, Ishowo-Adejumo R, Wolk R, Sadrarhami M, Lejeune A. Long-term safety and efficacy of ritlecitinib in adults and adolescents with alopecia areata and at least 25% scalp hair loss: Results from the ALLEGRO-LT phase 3, open-label study. J Eur Acad Dermatol Venereol. 2025 Jan 23. doi: 10.1111/jdv.20526. Epub ahead of print. PMID: 39846397.
Keywords:
Study, Highlights, Long-Term, Efficacy, Ritlecitinib, Alopecia Areata, Tziotzios C, Sinclair R, Lesiak A, Mehlis S, Kinoshita-Ise M, Tsianakas A, Luo X, Law EH, Ishowo-Adejumo R, Wolk R, Sadrarhami M, Lejeune A.
Dr. Shravani Dali has completed her BDS from Pravara institute of medical sciences, loni. Following which she extensively worked in the healthcare sector for 2+ years. She has been actively involved in writing blogs in field of health and wellness. Currently she is pursuing her Masters of public health-health administration from Tata institute of social sciences. She can be contacted at editorial@medicaldialogues.in.


