- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Moxidectin and albendazole inferior to ivermectin and albendazole for Trichuris trichiura infection
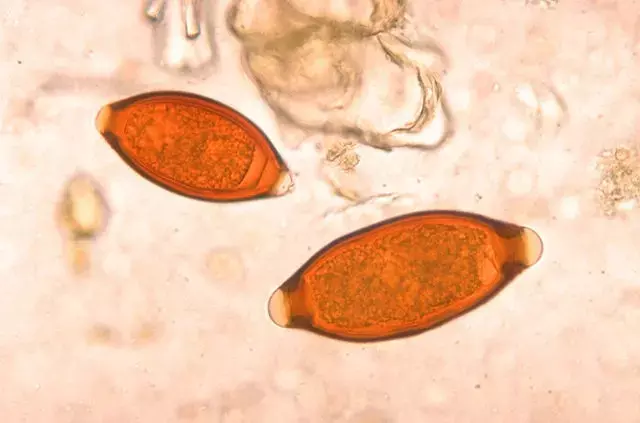
A recent study published in the Lancet shows inferiority of moxidectin and albendazole to ivermectin and albendazole against T trichiura.
Control efforts against soil-transmitted helminths focus on preventive chemotherapy with albendazole and mebendazole, however these drugs yield unsatisfactory results against Trichuris trichiura infections. We aimed to assess the efficacy and safety of moxidectin and albendazole compared with ivermectin and albendazole against T trichiura in adolescents living on Pemba Island, Tanzania.
This open-label, non-inferiority, randomised, controlled, phase 2/3 trial was done in four secondary schools (Kilindi, Kwale, Ndagoni [Chake Chake District], and Kiuyu [Wete District]) on Pemba Island, Tanzania. Adolescents aged 12–19 years who tested positive for T trichiura in at least two of four Kato-Katz slides with a mean infection intensity of 48 eggs per gram (EPG) of stool or higher were considered for inclusion. Participants were randomly assigned (21:21:2:2:8) to five treatment groups (8 mg moxidectin and 400 mg albendazole [group 1], 200 μg/kg ivermectin and 400 mg albendazole [group 2], 400 mg albendazole [group 3], 200 μg/kg ivermectin [group 4], or 8 mg moxidectin [group 5]) using a computer-generated randomisation code, stratified by baseline T trichiura infection intensity. Study site investigators and participants were not masked to study treatment; however, allocation was concealed to participants. The primary outcome was egg reduction rate (ERR) of T trichiura 14–21 days after treatment in the available case population. Moxidectin and albendazole was considered non-inferior to ivermectin and albendazole (control group) when the lower limit of the two-sided 95% CI of the difference was higher than the non-inferiority margin of –2 percentage points. This study is registered with ClinicalTrials.gov, NCT04700423.
Findings:
- Between March 1 and April 30, 2021, 771 participants were assessed for eligibility. 221 (29%) of 771 participants were ineligible and a further 14 (2%) were excluded. 207 (39%) of 536 participants were randomly assigned to moxidectin and albendazole, 211 (39%) to ivermectin and albendazole, 19 (4%) to albendazole, 19 (4%) to ivermectin, and 80 (15%) to moxidectin.
- Primary outcome data were available for all 536 participants.
- The geometric mean ERR of T trichiura after 14–21 days was 96·8% with moxidectin and albendazole and 99·0% with ivermectin and albendazole
- No serious adverse events were reported during the study.
- The most reported adverse events were headache (160 [34%] of 465), abdominal pain (78 [17%]), itching (44 [9%]), and dizziness (26 [6%]).
The findings show inferiority of moxidectin and albendazole to ivermectin and albendazole against T trichiura. However, given the high efficacy, moxidectin coadministration might complement treatment progammes, particularly in areas in which ivermectin is not available
Reference:
Efficacy and safety of moxidectin and albendazole compared with ivermectin and albendazole coadministration in adolescents infected with Trichuris trichiura in Tanzania: an open-label, non-inferiority, randomised, controlled, phase 2/3 trial
Dr. Shravani Dali has completed her BDS from Pravara institute of medical sciences, loni. Following which she extensively worked in the healthcare sector for 2+ years. She has been actively involved in writing blogs in field of health and wellness. Currently she is pursuing her Masters of public health-health administration from Tata institute of social sciences. She can be contacted at editorial@medicaldialogues.in.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


