- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
COVID-19 vaccine found to be 90% effective: Pfizer
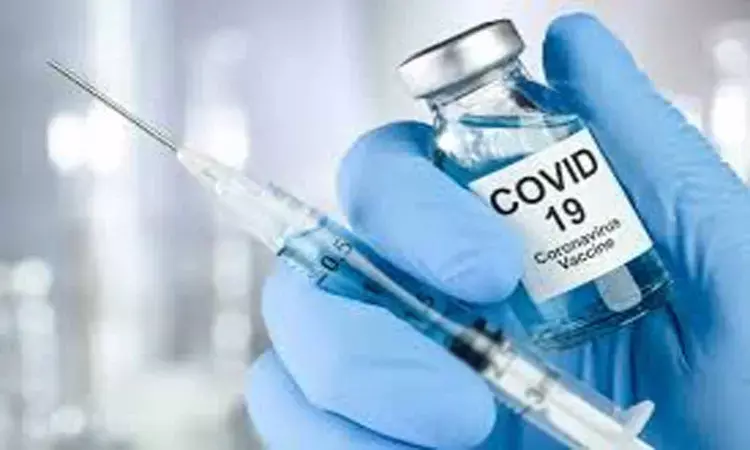
According to a recent interim analysis of a phase 3 study, a vaccine for SARS-CoV-2 shows 90% efficacy in fighting the virus when experimented on trial volunteers who were never exposed to the infection of the virus. Pfizer and BioNTech SE of the Trump administration's Operation Warp Speed is independently developing a messenger RNA–based vaccine by the name BTN162b2.
The vaccine requires two doses. The phase 3 trial has already enrolled a total of 43,538 participants among which a sparse 42% of enrollees have racially and ethnically diverse backgrounds. In their press release, Pfizer and BioNTech SE announced that a sample of 38,955 trial volunteers received a second dose of either vaccine or placebo. Out of the total participants, an interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine showed above 90% efficacy rate, 7 days postoperatively after the second dose. This illustrated the fact that protection against the virus was achieved 28 days after the first vaccine dose. The companies will tabulate a final data analysis after the 164 confirmed COVID-19 cases have occurred. So far, there have not been any serious complications noted by the DMC. "It's promising in that it validates the genetic strategy – whether its mRNA vaccines or DNA vaccines," Paul A. Offit, MD, and member of the US Food and Drug Administration's COVID-19 Vaccine Advisory Committee told Medscape Medical News. "All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it's effective. That's good. We knew that it seemed to work in experimental animals, but you never know until you put it into people", he further added. Pfizer CEO Albert Bourla, DVM, Ph.D., added in a separate press release, "It's important to note that we
Dr. Nandita Mohan is a practicing pediatric dentist with more than 5 years of clinical work experience. Along with this, she is equally interested in keeping herself up to date about the latest developments in the field of medicine and dentistry which is the driving force for her to be in association with Medical Dialogues. She also has her name attached with many publications; both national and international. She has pursued her BDS from Rajiv Gandhi University of Health Sciences, Bangalore and later went to enter her dream specialty (MDS) in the Department of Pedodontics and Preventive Dentistry from Pt. B.D. Sharma University of Health Sciences. Through all the years of experience, her core interest in learning something new has never stopped. She can be contacted at editorial@medicaldialogues.in. Contact no. 011-43720751
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


