- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Covid-19: Sputnik V due to submit vaccine data to WHO by December end
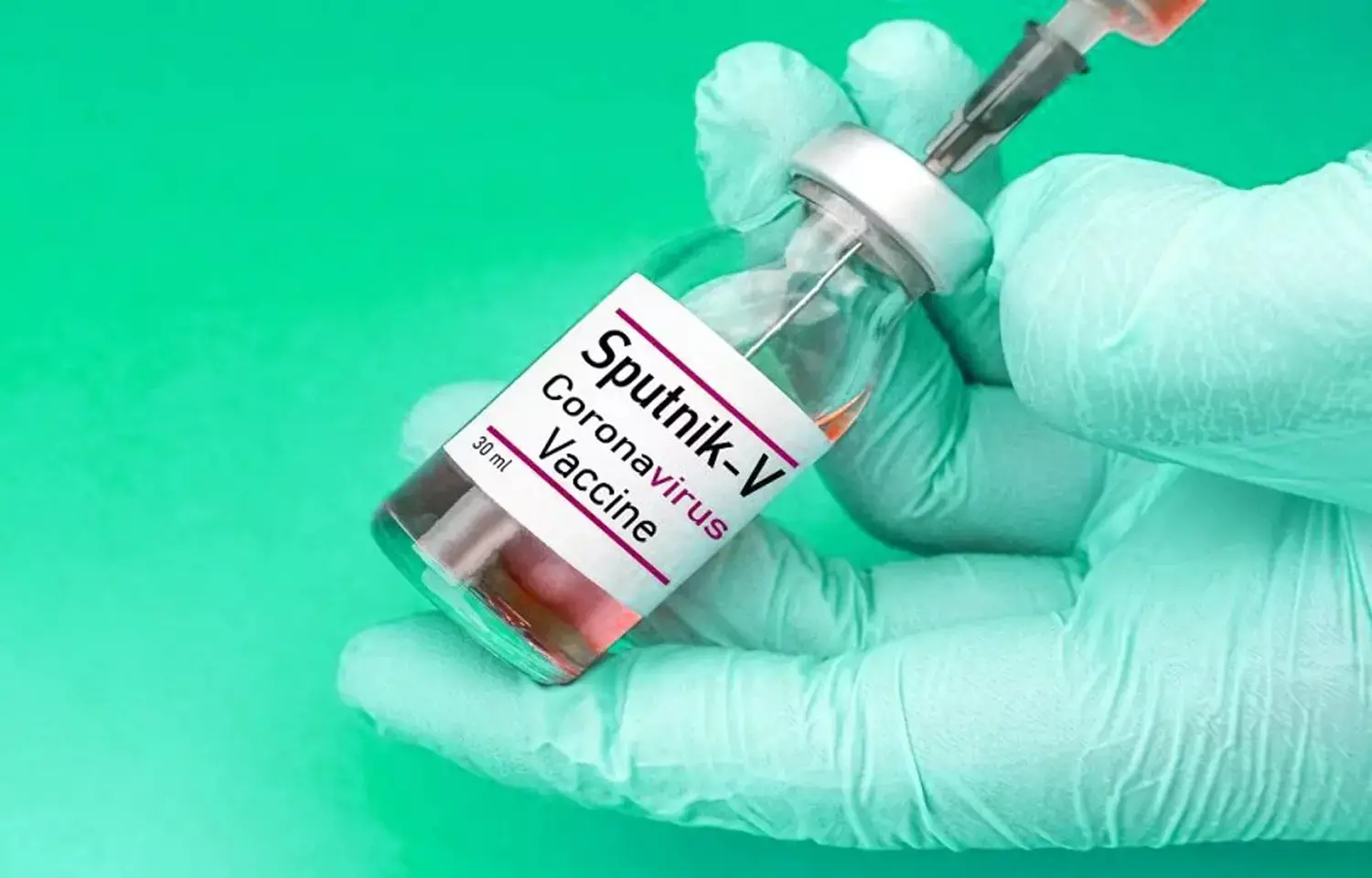
Geneva: The Russian maker of the COVID-19 Sputnik V vaccine is due to submit its latest clinical data by the end of December, with manufacturing site inspections expected to follow in February, a World Health Organization (WHO) official said on Monday.
Moscow rushed to approve the shot for domestic use last year and it has been exported to countries around the world, but it has still not been certified by either the WHO or the European Medicines Agency, the EU's drug regulator.
Rogerio Gaspar, WHO regulation director, gave the new timelines for the vaccine made by the Gamaleya Institute, which is seeking WHO emergency use listing, during a WHO briefing for journalists in Geneva.
It was hoped that Gamaleya would complete submission of its data and application in two parts, at the end of December and by the end of January, he said.
"We are starting already for the planning exercise for the forthcoming GMP (good manufacturing practices) inspections that will be necessary looking at the new data to be submitted," Gaspar said, referring to on-site inspections for quality control.
"So our planning right now, provided that all information is on board and with the technical information being answered by the end of December, we would be able to perform GMP (good manufacturing practices) inspections locally on Sputnik in February," he said.
The Sputnik V vaccine is based on a proven well-studied human adenovirus vector platform; these vectors cause the common cold and have plagued humanity for millennia.
In the past year, the WHO has approved nine COVID-19 vaccines for emergency use listing, which assesses their quality, safety and efficacy and is a prerequisite for providing doses to the COVAX vaccine supply for poorer countries.
The listing also allows countries to expedite their own regulatory approval to import and administer COVID-19 vaccines, reports Reuters.
Ruchika Sharma joined Medical Dialogue as an Correspondent for the Business Section in 2019. She covers all the updates in the Pharmaceutical field, Policy, Insurance, Business Healthcare, Medical News, Health News, Pharma News, Healthcare and Investment. She has completed her B.Com from Delhi University and then pursued postgraduation in M.Com. She can be contacted at editorial@medicaldialogues.in Contact no. 011-43720751
Next Story


