- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
DCGI gives nod to Bharat Biotech Intranasal 'Five Arms' COVID booster dose for restricted use
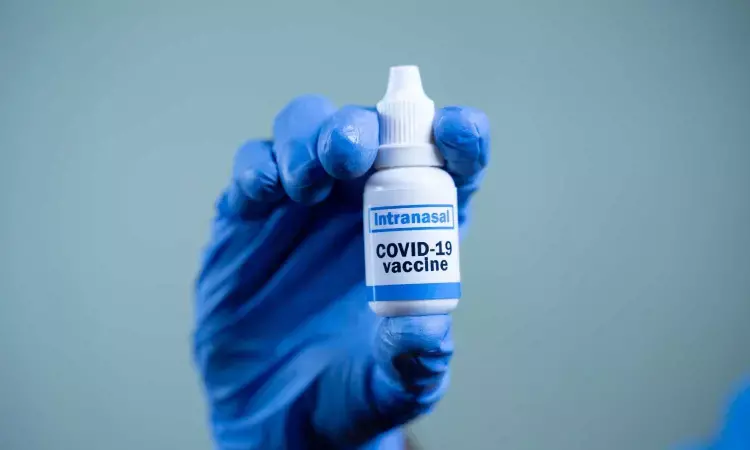
The nasal vaccine can be taken after six months of the second dose. As this is non-invasive and needle-free, it becomes easy to administer.
New Delhi: Vaccine manufacturer Bharat Biotech has received approval from the Drugs Controller General of India (DCGI) for an Intranasal 'Five Arms' Covid-19 booster dose for restricted use.
A source said that the emergency use authorisation (EUA) has been given to the nasal vaccine iNCOVACC for restricted emergency use as the third dose for adults irrespective of having been administered Covaxin or Covishield vaccine doses.
BBV154 - A novel adenovirus vectored, intranasal vaccine for COVID-19.
As per the vaccine manufacturer, the nasal route has excellent potential for vaccination due to the organised immune systems of the nasal mucosa. Thus, intranasal immunisation of ChAd-SARS-CoV-2-S can create an immune response in the nose, which is the point of entry for the virus -- thereby protecting against disease, infection, and transmission.
The nasal vaccine can be taken after six months of the second dose. As this is non-invasive and needle-free, it becomes easy to administer.
Bharat Biotech claimed that the intranasal vaccine stimulates a broad immune response. It is likely to block both infection and transmission of COVID-19.
Coronavirus disease (COVID-19) is an infectious disease caused by the SARS-CoV-2 virus.
According to WHO, most people infected with the virus will experience mild to moderate respiratory illness and recover without requiring special treatment. However, some will become seriously ill and require medical attention. Older people and those with underlying medical conditions like cardiovascular disease, diabetes, chronic respiratory disease, or cancer are more likely to develop serious illness. Anyone can get sick with COVID-19 and become seriously ill or die at any age.
Read also: DCGI Panel To Hold Discussion On Intranasal "Five Arms" Booster Dose INCOVACC On Nov 15: Sources
Bharat Biotech International Limited is an Indian multinational biotechnology company headquartered in Genome Valley, Turakapally, Hyderabad. The company was founded by Krishna Ella in 1996.
It is engaged in drug discovery, drug development, manufacture of vaccines, bio-therapeutics, pharmaceuticals, and health care products.
Ruchika Sharma joined Medical Dialogue as an Correspondent for the Business Section in 2019. She covers all the updates in the Pharmaceutical field, Policy, Insurance, Business Healthcare, Medical News, Health News, Pharma News, Healthcare and Investment. She has completed her B.Com from Delhi University and then pursued postgraduation in M.Com. She can be contacted at editorial@medicaldialogues.in Contact no. 011-43720751


