- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
USFDA approves Alembic Pharma Icatibant Injection for acute attacks of hereditary angioedema
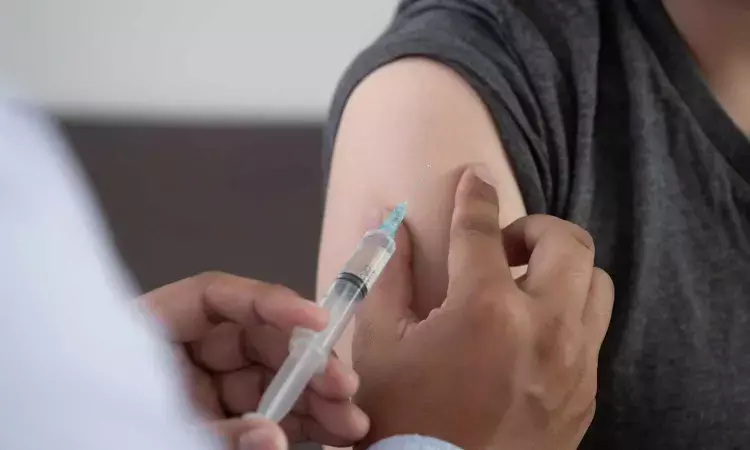
Icatibant injection is indicated for the treatment of acute attacks of hereditary angioedema in adults 18 years of age and older.
Vadodara: Alembic Pharmaceuticals Limited today announced that it has received final approval from the US Food & Drug Administration (USFDA) for its Abbreviated New Drug Application (ANDA) for Icatibant Injection, 30 mg/3 mL (10 mg/mL) Single-Dose Prefilled Syringe.
The approved ANDA is therapeutically equivalent to the reference listed drug product (RLD), Firazyr Injection, 30 mg/3 mL (10 mg/mL), of Takeda Pharmaceuticals U.S.A., Inc.
Icatibant injection is indicated for the treatment of acute attacks of hereditary angioedema (HAE) in adults 18 years of age and older.
Hereditary angioedema is a rare genetic disorder characterized by recurrent episodes of swelling (edema) in various parts of the body, including the face, limbs, abdomen, and throat. These episodes are caused by a deficiency or dysfunction of C1 esterase inhibitor, a protein involved in regulating the immune response and inflammation. HAE attacks can be triggered by stress, trauma, or certain medications, and they typically last for several days
This is the first peptide product approval from the USFDA received by the Company. Icatibant Injection has an estimated market size of US$ 112 million for twelve months ending Mar 2024 according to IQVIA. Alembic has a cumulative total of 205 ANDA approvals (177 final approvals and 28 tentative approvals) from USFDA.
Read also: Alembic Pharma gets USFDA nod for Sacubitril and Valsartan Tablets for heart failure
Alembic Pharmaceuticals Limited is a vertically integrated research and development pharmaceutical company. Headquartered in India, Alembic is a publicly listed company that manufactures and markets generic pharmaceutical products all over the world. Alembic's state of the art research and manufacturing facilities are approved by regulatory authorities of many developed countries including the USFDA.
Read also: USFDA approves Alembic Pharma five products in over a month
Ruchika Sharma joined Medical Dialogue as an Correspondent for the Business Section in 2019. She covers all the updates in the Pharmaceutical field, Policy, Insurance, Business Healthcare, Medical News, Health News, Pharma News, Healthcare and Investment. She has completed her B.Com from Delhi University and then pursued postgraduation in M.Com. She can be contacted at editorial@medicaldialogues.in Contact no. 011-43720751
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


