- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Novel EpiFaith syringe for epidural anesthesia bests regular loss-of-resistance syringe: IJOA
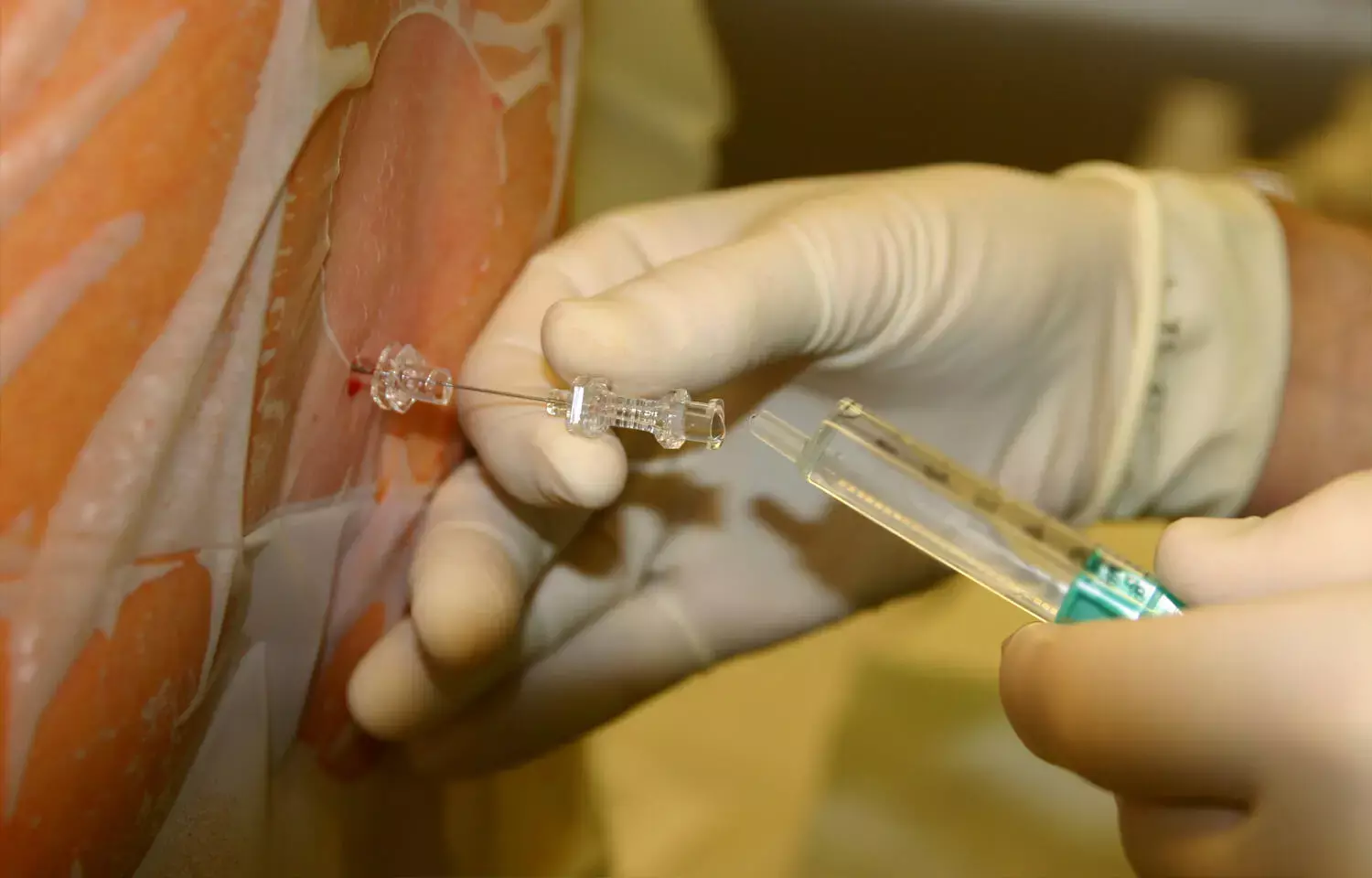
The EpiFaith syringe (Flat Medical, Taipei City, Taiwan) is a new, spring-loaded loss-of-resistance (LOR) syringe designed to aid locating the tip of a Tuohy needle in the epidural space. Compared to the standard LOR technique with a non‐spring‐loaded syringe in which the operator's thumb is used to provide force to the syringe plunger, the EpiFaith® provides an autonomous and objective indication of the LOR that is similar to another product, the Episure® Autodetect® LOR syringe (Indigo Orb Inc., Santa Clara, CA, USA). Both syringes use spring‐loaded plungers, however the EpiFaith® has the ability to provide a variable load to the spring. Additionally, the EpiFaith® is not loaded until the Tuohy needle tip is sited in the interspinous ligament. This simplifies its use. The syringe has been studied previously only on animal and phantom models.
M.W. Athar et al. in his study evaluated the EpiFaith® syringe in a clinical setting, among a cohort of pregnant women receiving neuraxial labor analgesia.
In this non‐randomized observational study, four anesthesiologists used the EpiFaith® syringe 10 times each while placing epidural catheters for labor analgesia. The anesthesiologists scored each placement on an 11‐point Likert scale (−5 = absolutely worse, 0 = the same, and 5 = absolutely better than using their regular loss‐of‐resistance syringe technique).
All procedures were performed with the patient in the sitting position. A 17‐gauge Perifix® Tuohy needle was inserted at the L3/4 interspace until the anesthesiologist felt the Tuohy needle had passed through the supraspinous ligament and was engaged in the interspinous ligament. The EpiFaith® syringe, loaded with air or saline, was then attached to the Tuohy needle's hub. Successful epidural catheter placement was assumed if two consecutive pain scores of <3/10 were attained within 45 min of block placement.
The primary outcome was correctly locating the epidural space (defined as both LOR and obtaining cerebrospinal fluid at the CSE or DPE technique).
- In 90% of the cases, a clear LOR endpoint was reported by the anesthesiologists. All 40 neuraxial placements obtained cerebrospinal fluid during the spinal component of the CSE or DPE techniques, and 39 of 40 epidural catheters provided adequate labor analgesia.
- Air was used in the EpiFaith® syringe to detect LOR in 35 cases and saline in five cases.
- In 50%, 27.5% and 22.5% of cases, respectively, the anesthesiologist reported that using the EpiFaith® syringe was better than, the same as, or worse than using their regular LOR syringe.
- The median Likert scale score comparing the EpiFaith® syringe with the anesthesiologist's regular LOR syringe was >0 for three of the four anesthesiologists. Anesthesiologists #2 and #3 had scored the use of the EpiFaith® syringe significantly higher than anesthesiologist #4 (P<0.001 in both cases). There was no trend up or down in the Likert scale scores with more experience using the syringe (P=0.54).
- In eight cases, the anesthesiologists required more than one pass. In four cases, the tension of the syringe spring released before reaching the epidural space, and the EpiFaith® syringe had to be re‐loaded. The reported leakage in these cases was slow and with slight movement of the syringe plunger, as opposed to the rapid movement of the syringe plunger when the Tuohy needle tip reached the epidural space. Anesthesiologists mentioned two cases in which there was a brief delay in the spring release when they entered the epidural space. One anesthesiologist changed to their regular LOR syringe on one occasion. There were no unintentional dural punctures with the Tuohy needle and no postural headaches identified on follow‐up.
This feasibility study showed that the EpiFaith® syringe reliably detected the epidural space. Three of the four operators scored it favorably compared with their usual syringe. Authors hypothesized that this is due to the EpiFaith® syringe allowing the anesthesiologist to use both hands to control the Tuohy needle and focus solely on the needle's advancement speed and trajectory.
Although this study was underpowered to evaluate complication outcomes, it was encouraging that there were no unintentional dural punctures or postpartum postural headaches associated with the use of the EpiFaith® syringe.
The EpiFaith® syringe is potentially a useful learning tool in teaching the epidural LOR technique to trainees because the visual sign of epidural placement removes operator subjectivity. A similar spring‐ loaded LOR syringe (Episure®) showed a higher success rate than a standard LOR syringe for identification of the epidural space in parturient.
In this study the Likert‐scaled scores and time for placement did not change with the number of times the syringe was used by the anesthesiologist. This is probably because the device does not have a steep learning curve.
The manufacturer suggested that both air and saline can be used within the EpiFaith® syringe. When practising with the syringe on a phantom model prior to the clinical trial, authors concluded that the syringe plunger was more responsive when used with air. The anesthesiologist who used saline in 5 out of 10 attempts for the reason of comparison found that the syringe plunger's responsiveness aligned with what authors had noted in the phantom model. If saline is used, authors recommend that the spring is loaded to the maximum setting in order to improve the plunger's responsiveness.
The authors concluded, "This feasibility study found that the EpiFaith® syringe reliably detected the epidural space. Three of the four anesthesiologists in this trial rated their experience with the EpiFaith® syringe as equal to, or better than, using their usual LOR syringe in 78% of the insertions, and there were no reported complications. The results of this initial study to evaluate the EpiFaith® syringe clinically suggest a potential role for this novel device to aid epidural catheter placement. Future large randomized studies are required to determine if outcomes such as the incidence of failed neuraxial blockade and unintentional dural puncture will be improved by the use of this LOR syringe."
Source: M.W. Athar et al; International Journal of Obstetric Anesthesia 47 (2021)
https://doi.org/10.1016/j.ijoa.2021.102984
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


