- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Topical Cyclosporine A cationic ophthalmic emulsion effective treatment option for vernal keratoconjunctivitis in kids
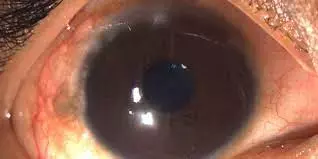
Topical Cyclosporine A cationic ophthalmic emulsion effective treatment option for vernal keratoconjunctivitis in kids suggests a new study published in the Eye
Cyclosporine A cationic ophthalmic emulsion (CsA CE) was evaluated in paediatric and adolescent patients with vernal keratoconjunctivitis (VKC) in the NOVATIVE (NCT00328653) and VEKTIS (NCT01751126) trials. The similarity of these studies permitted pooled assessment of the effect of Cyclosporine A cationic ophthalmic emulsion on corneal damage as well as safety and tolerability.
Subjects/Methods
Pooled outcomes were assessed for the first 28 days of treatment. In NOVATIVE, 118 patients were randomised to 4 times daily (QID) Cyclosporine A cationic ophthalmic emulsion 0.05%, 0.1%, or vehicle eye drops. In VEKTIS, 169 patients were randomised to Cyclosporine A cationic ophthalmic emulsion 0.1% QID or twice daily (BID) or vehicle. For these analyses, treatment groups comprised: (1) pooled CsA CE 0.1% QID arms (high-dose; n = 96); (2) pooled CsA CE 0.05% QID arm from NOVATIVE and Cyclosporine A cationic ophthalmic emulsion 0.1% BID data from VEKTIS (low-dose; n = 93); and (3) pooled vehicle QID arms (vehicle; n = 98).
Results
Changes from baseline to day 28 (mean ± standard deviation) in corneal fluorescein staining (CFS) scores for Cyclosporine A cationic ophthalmic emulsion high-dose, low-dose, and vehicle groups were −1.6 ± 1.47 (95% CI: −0.9, −0.1; p = 0.0124 vs vehicle), −1.7 ± 1.39 (95% CI: −1.1, −0.3; p = 0.0015 vs vehicle), and −1.0 ± 1.55, respectively. Adverse events (AEs) of any type were reported in 37.5%, 34.4%, and 37.8% of the high-dose, low-dose, and vehicle groups, respectively. Most were mild or moderate in severity.
Cyclosporine A cationic ophthalmic emulsion significantly decreased corneal damage and was safe and well tolerated in patients with VKC. These data support Cyclosporine A cationic ophthalmic emulsion as a treatment option for the management of VKC.
Reference:
Leonardi, A., Doan, S., Aragona, P. et al. Topical cyclosporine A cationic ophthalmic emulsion in paediatric vernal keratoconjunctivitis: pooled analysis of randomised NOVATIVE and VEKTIS trials. Eye 37, 2320–2326 (2023). https://doi.org/10.1038/s41433-022-02342-6
Dr. Shravani Dali has completed her BDS from Pravara institute of medical sciences, loni. Following which she extensively worked in the healthcare sector for 2+ years. She has been actively involved in writing blogs in field of health and wellness. Currently she is pursuing her Masters of public health-health administration from Tata institute of social sciences. She can be contacted at editorial@medicaldialogues.in.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


