- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
FDA approves wheeled CT scanner allowing bedside scanning in ICU
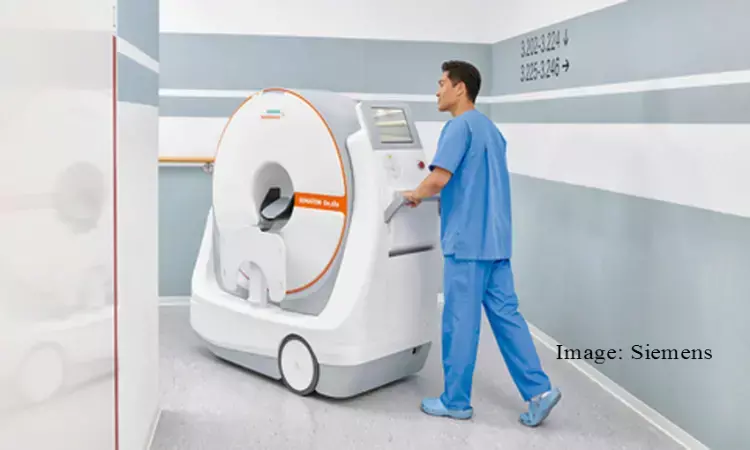
USA: The US Food and Drug Administration (FDA) has approved Siemens Healthineers' wheeled CT scanner. The Somatom scanner consists of motorized wheels and a forward-looking camera that allows a single person to navigate it through the hospital. This allows the convenience of bringing the scanner to the patient instead of the other way round, allowing the easier head exams of critically ill patients. This eliminates the risks and logistics of transporting the patient through the hospital to a radiology unit to use a larger machine.
The scanner includes the necessary computers and batteries, as well as keyboard and touchscreen for operation inside smaller rooms without requiring extra equipment. After the system is locked in place at the end of a patient's bed, the scanner moves on a telescopic gantry to acquire the image, with the person's head resting within a 35 cm bore.
Read Also: Good or Bad: CT has decreased but pediatric advanced imaging has increased
"The Somatom On.site is a fundamentally new approach for CT head scans of intensive care patients," Siemens head of computed tomography, Philipp Fischer, said late last year after the device debuted at the December 2019 assembly of the Radiological Society of North America.
"At the same time, it allows healthcare providers to use their staff and their permanently installed CT fleet even more effectively," Fischer said, with ICU patients typically requiring more time to be safely moved through a radiology suite compared to more conventional exams.
In addition, the scanner also features radiation curtain shields that can be attached to the front and rear, allowing clinicians to stay nearby during scanning, while the patient can remain connected to any monitors and devices.
Read Also: Cardiac CT an opportunistic but effective way to screen osteoporosis
Mobile CT scanner reimagines head imaging of critically ill patients by enabling patients and staff to remain in ICU.
MSc. Biotechnology
Medha Baranwal holds a Bachelor’s degree in Biomedical Sciences from the University of Delhi and a Master’s degree in Biotechnology from Amity University. Since May 2018, she has been contributing to Medical Dialogues, writing and editing medical news articles that translate complex research into clear, accessible information for healthcare professionals.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


