- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Increased PSMA PET/CT may predict chemotherapy response in metastatic prostate cancer: Study
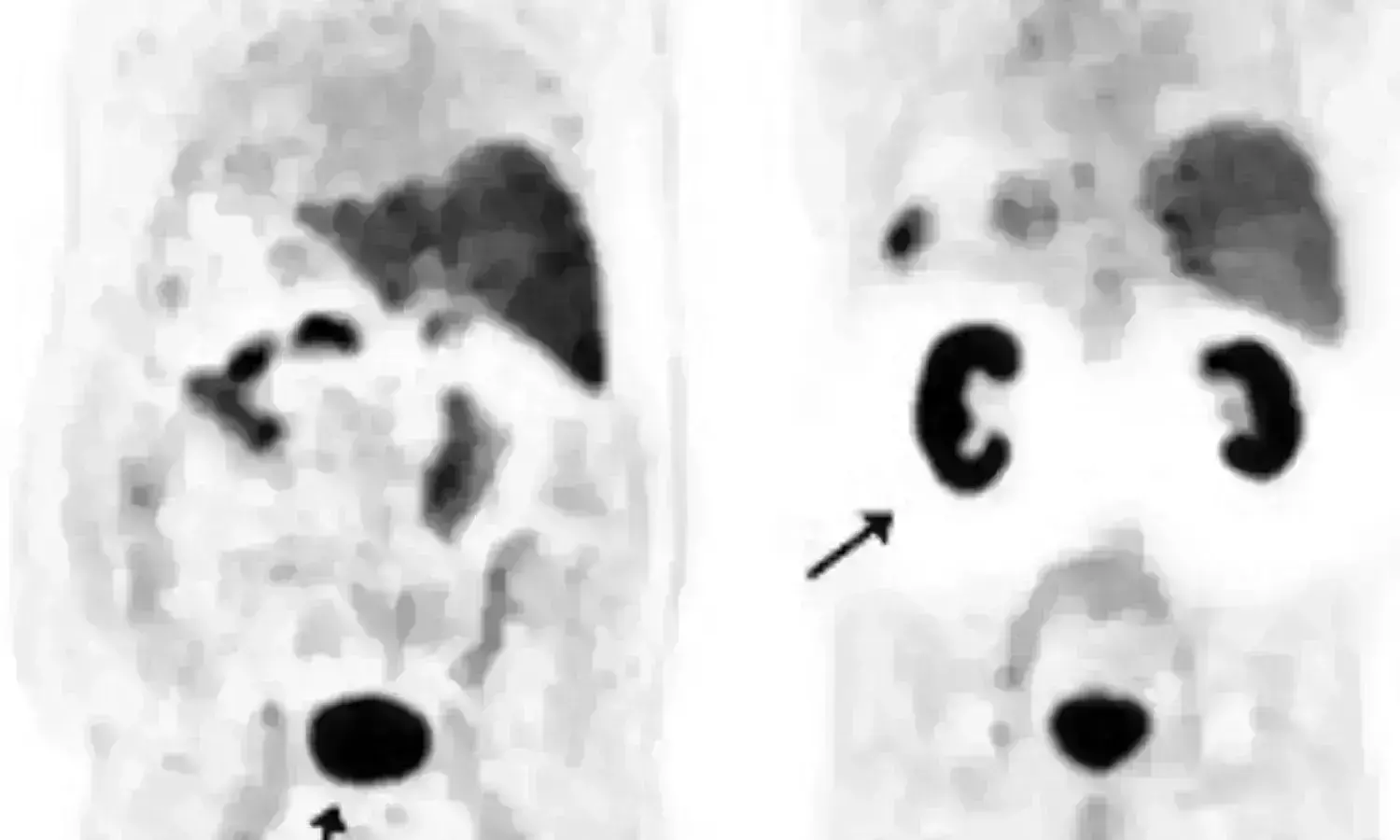
Belgium: 68Ga-PSMA-11 PET/CT can help predict if patients with metastatic prostate cancer (mPCa) will respond to taxane-based chemotherapy, suggests results from a retrospective analysis. Prostate-specific membrane antigen (PSMA)-expression changes can be used as a predictive biomarker for overall survival (OS) which might be helpful for customizing individual therapy and selecting patients eligible for clinical trials. The study was published in the Journal of Nuclear Medicine on November 12, 2021.
Qaid Ahmed Shagera, Department of Nuclear Medicine, Institut Jules Bordet, Universite Libre de Bruxelles (ULB), Belgium, and colleagues aimed to evaluate the role of Positron Emission Tomography (PET) targeting the PSMA for response assessment in mPCa treated with taxane-based chemotherapy (docetaxel or cabazitaxel) and its predictive value on patient outcome.
For this purpose, the researchers retrospectively evaluated 37 patients with metastatic hormone-sensitive or castration-resistant prostate cancer (mHSPC or mCRPC) who underwent t 68Ga-PSMA-11 PET/CT at baseline and after the last cycle of taxane-based chemotherapy (docetaxel or cabazitaxel) without treatment modification between scans.
Biochemical response (BR) was defined as an undetectable or decreased prostate-specific antigen (PSA) by ≥50% compared to baseline. They also tested the association between BR and different PET parameters. A cut-off of ≥30% change in PSMA total tumor volume (PSMA-TV) was used to define PSMA responders (PSMA-R) vs PSMA non-responders (PSMA-NR).
The study cohort was composed of 8 (22%) mHSPC and 29 (78%) mCRPC patients. Twenty-one patients received docetaxel, and 16 received cabazitaxel treatment.
Based on the study, the researchers found the following:
- BR was found in 18/37 patients.
- Using PSMA-TV, PSMA-PET/CT response was concordant with BR in 35/37 patients (Phi=0.89).
- There were 18/37 PSMA-R (6 complete response and 12 partial response) and 19/37 PSMA-NR (17 progressive disease and 2 stable disease).
- After a median follow-up of 23 months there was a statistically significant longer overall survival (OS) for PSMA-R compared to PSMA-NR (median OS not reached vs 12 months, respectively, HR 0.10) for the entire population.
- Among the mCRPC subgroup, differences in OS were also observed (median 22 vs 12 months respectively, HR 0.22) with a 12-month OS rate of 100% for PSMA-R and 52% for PSMA-NR.
"This retrospective analysis suggests that 68Ga-PSMA-11 PET/CT is a promising imaging modality for assessing response to taxane-based chemotherapy in mPCa," wrote the authors. "PSMA-expression changes might be used as a predictive biomarker for OS which might help tailor individual therapy and select eligible patients for clinical trials."
Reference:
68Ga-PSMA PET/CT for response assessment and outcome prediction in metastatic prostate cancer patients treated with taxane-based chemotherapy. Qaid Ahmed Shagera, Carlos Artigas, Ioannis Karfis, Gabriela Critchi, Nieves Martinez Chanza, Spyridon Sideris, Alexandre Peltier, Marianne Paesmans, Thierry Gil, Patrick Flamen. Journal of Nuclear Medicine Nov 2021, jnumed.121.263006; DOI: 10.2967/jnumed.121.263006
MSc. Biotechnology
Medha Baranwal holds a Bachelor’s degree in Biomedical Sciences from the University of Delhi and a Master’s degree in Biotechnology from Amity University. Since May 2018, she has been contributing to Medical Dialogues, writing and editing medical news articles that translate complex research into clear, accessible information for healthcare professionals.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


