- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Scientists transplant two gene edited pig kidneys into brain-dead human
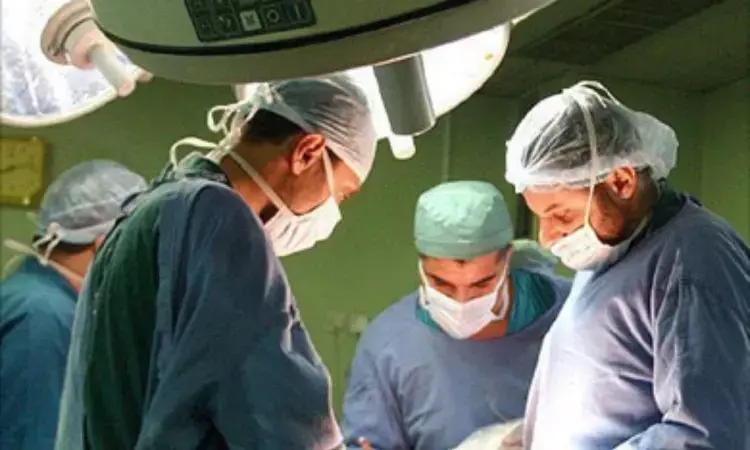
BIRMINGHAM, Ala. - The University of Alabama at Birmingham Marnix E. Heersink School of Medicine have successfully transplanted two kidneys from a genetically modified pig into a brain dead human recipient. Further the transplaned kidneys produced urine and were not rejected during the days-long experiment.
They have announced the first peer-reviewed research outlining the successful transplant of genetically modified, clinical-grade pig kidneys into a brain-dead human individual, replacing the recipient's native kidneys. These positive results demonstrate how xenotransplantation could address the worldwide organ shortage crisis.
In the study published in the American Journal of Transplantation, UAB researchers tested the first human preclinical model for transplanting genetically modified pig kidneys into humans. The study recipient had two genetically modified pig kidneys transplanted in his abdomen after his native kidneys were removed. The organs were procured from a genetically modified pig at a pathogen-free facility.
"Along with our partners, we have made significant investments in xenotransplantation for almost a decade hoping for the kinds of results published today," said Selwyn Vickers, M.D., dean of the UAB Heersink School of Medicine and CEO of the UAB Health System and UAB/Ascension St. Vincent's Alliance. "Today's results are a remarkable achievement for humanity and advance xenotransplant into the clinical realm. With this study, our research teams have also demonstrated that the decedent model has significant potential to propel the xenotransplantation field forward."
For the first time, the pig kidneys transplanted were taken from pigs that had been genetically modified with 10 key gene edits that may make the kidneys suitable for transplant into humans. This process demonstrates the long-term viability of the procedure and how such a transplant might work in the real world. The transplanted kidneys filtered blood, produced urine and, importantly, were not immediately rejected. The kidneys remained viable until the study was ended, 77 hours after transplant.
"This game-changing moment in the history of medicine represents a paradigm shift and a major milestone in the field of xenotransplantation, which is arguably the best solution to the organ shortage crisis," said Jayme Locke, M.D., director of the Comprehensive Transplant Institute in UAB's Department of Surgery and lead surgeon for the study. "We have bridged critical knowledge gaps and obtained the safety and feasibility data necessary to begin a clinical trial in living humans with end-stage kidney failure disease."
Gene editing in pigs to reduce immune rejection has made organ transplants from pigs to humans possible, which could offer help to thousands of people who face organ failure, disease or injury. The natural lifespan of a pig is 30 years, they are easily bred and can have organs of similar size to humans.
Genetically modified pig kidneys have been extensively tested in non-human primates. In addition to testing in non-human primates, evaluating genetically modified pig kidneys in a human preclinical model research may provide important information about the potential safety and efficacy of kidneys in human transplant recipients, including in clinical trials.
"This human preclinical model is a way to evaluate the safety and feasibility of the pig-to-non-human primate model, without risk to a living human," Locke added. "Our study demonstrates that major barriers to human xenotransplantation have been surmounted, identifies where new knowledge is needed to optimize xenotransplantation outcomes in humans, and lays the foundation for the establishment of a novel preclinical human model for further study."
This effort is supported by biotechnology pioneer United Therapeutics Corporation, which awarded a grant to UAB to launch the innovative xenotransplantation program. Revivicor, Inc., a subsidiary of United Therapeutics, provided the genetically modified pig that was the source of the investigational xenotransplant kidneys called UKidney™.
"All of us at Revivicor are in awe of the historic achievements at UAB with our investigational 10-gene xenokidney, or UKidney," said David Ayares, Ph.D., Chief Scientific Officer of Revivicor and a trailblazing genetic engineer since his early work cloning the world's first pigs and the first alpha-Gal knockout pigs. "We feel confident that this UKidney may turn out to be a life-saving solution for thousands of people on dialysis, subject to successful completion of our clinical trials and achievement of FDA approval in the next several years."
Hina Zahid Joined Medical Dialogue in 2017 with a passion to work as a Reporter. She coordinates with various national and international journals and association and covers all the stories related to Medical guidelines, Medical Journals, rare medical surgeries as well as all the updates in the medical field. Email: editorial@medicaldialogues.in. Contact no. 011-43720751
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


