- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Colchicine, a potential first-line treatment for cutaneous polyarteritis nodosa: Study
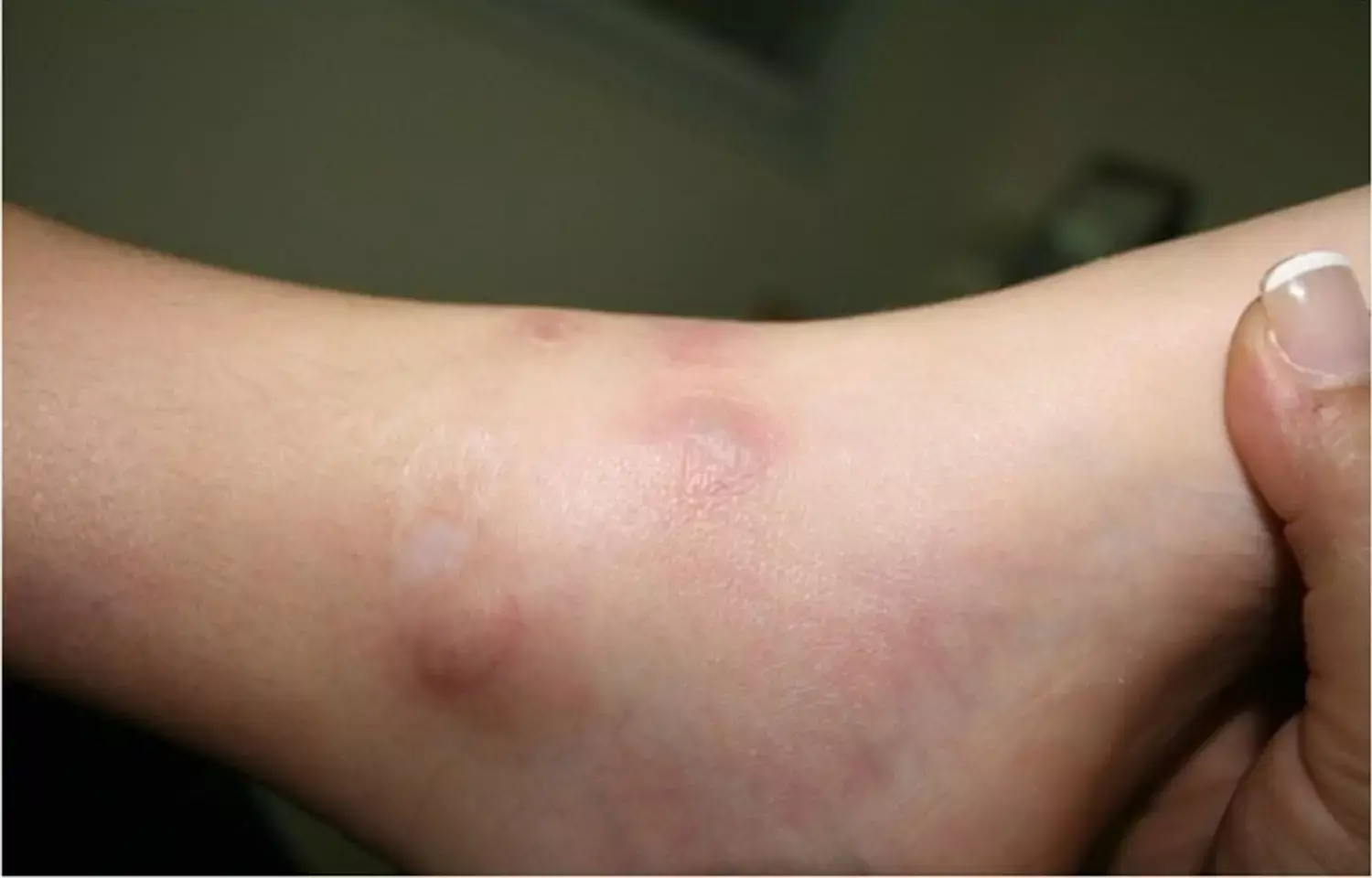
Paris: Colchicine is a reasonable first-line treatment for cutaneous polyarteritis nodosa (cPAN) without peripheral sensory neuropathy and has a favorable risk-to-benefit ratio, a recent study has found.
The study, published in the Journal of the American Academy of Dermatology, suggests the use of glucocorticoids (GCs) + azathioprine for patients with sensory neuropathy.
Cutaneous polyarteritis nodosa (cPAN) is a form of medium-sized vessel vasculitis. Despite it being a disabling and prolonged course, there is a lack of data on treatment efficacy and safety. To fill this knowledge gap, Alexis Régent, Université de Paris, Paris, and colleagues aimed to describe treatment efficacy and safety in patients with cPAN.
For this purpose, the researchers conducted a multicentre retrospective observational study. They recorded clinical and biological data together with treatments received. The primary outcome was the rate of complete response (CR) at month 3. Secondary outcomes included drug survival, and safety was assessed.
A total of 68 patients who received a median of 2 therapeutic lines (interquartile range 1-3) were included.
The research yielded the following findings:
- Overall, 31% patients achieved CR with colchicine, 23% with dapsone, 44% with glucocorticoids (GCs) alone, 11% with NSAIDs, 84% with GCs+azathioprine (AZA) and 47% with GCs+methotrexate.
- GCs+AZA had the best drug survival (median duration 29.5 months).
- Response at month 3 was decreased with peripheral neurological involvement (odds ratio 0.19).
- Overall, the rate of treatment-related adverse events was 18%, which led to treatment discontinuation in 7% of patients.
"Colchicine seems to confer good benefit–risk balance in cPAN without peripheral sensory neuropathy," wrote the authors. "GCs+AZA seems the best treatment for disease relapse."
Reference:
The study titled, "Efficacy and safety of treatments in cutaneous polyarteritis nodosa: a French observational retrospective study," is published in the Journal of the American Academy of Dermatology.
DOI: https://www.jaad.org/article/S0190-9622(21)02021-1/fulltext
MSc. Biotechnology
Medha Baranwal holds a Bachelor’s degree in Biomedical Sciences from the University of Delhi and a Master’s degree in Biotechnology from Amity University. Since May 2018, she has been contributing to Medical Dialogues, writing and editing medical news articles that translate complex research into clear, accessible information for healthcare professionals.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751